Carbon dioxide (CO2) is a naturally occurring molecule that plays a vital role in Earth's atmosphere and various biological processes. Understanding its molecular shape is essential for comprehending its chemical properties and behavior. This article delves into the molecular geometry of carbon dioxide, its significance, and related concepts.
Carbon dioxide is a fascinating molecule due to its unique structure and properties. It is a critical component of the Earth's atmosphere and plays a pivotal role in global climate systems. As we explore the molecular shape of carbon dioxide, we will uncover why this molecule has a linear geometry and how it impacts its reactivity and interactions.
In this guide, we will provide an in-depth analysis of the molecular shape of carbon dioxide, its chemical properties, and its implications. Whether you're a student, researcher, or simply curious about chemistry, this article aims to answer your questions comprehensively.
Introduction to Carbon Dioxide
Carbon dioxide (CO2) is a simple molecule composed of one carbon atom and two oxygen atoms. It is a colorless and odorless gas that exists naturally in the Earth's atmosphere. CO2 is a byproduct of respiration, combustion, and volcanic activity.
Why Study Carbon Dioxide?
Carbon dioxide is not only a critical component of the atmosphere but also a molecule of great interest in chemistry. Its molecular shape determines many of its physical and chemical properties, making it an essential subject of study for scientists.
The molecular geometry of CO2 is linear, which significantly influences its behavior in various chemical reactions. Understanding this shape helps explain why CO2 is nonpolar and how it interacts with other molecules.
What is Molecular Shape?
Molecular shape refers to the three-dimensional arrangement of atoms in a molecule. This geometry is determined by the valence shell electron pair repulsion (VSEPR) theory, which predicts the spatial arrangement of electron pairs around a central atom.
Importance of Molecular Shape
The molecular shape of a compound dictates its chemical properties, reactivity, and interactions with other substances. For instance, the shape of a molecule can determine whether it is polar or nonpolar, influencing its solubility and reactivity.
In the case of carbon dioxide, its linear shape plays a crucial role in its nonpolar nature and its ability to dissolve in water, forming carbonic acid.
Molecular Geometry of Carbon Dioxide
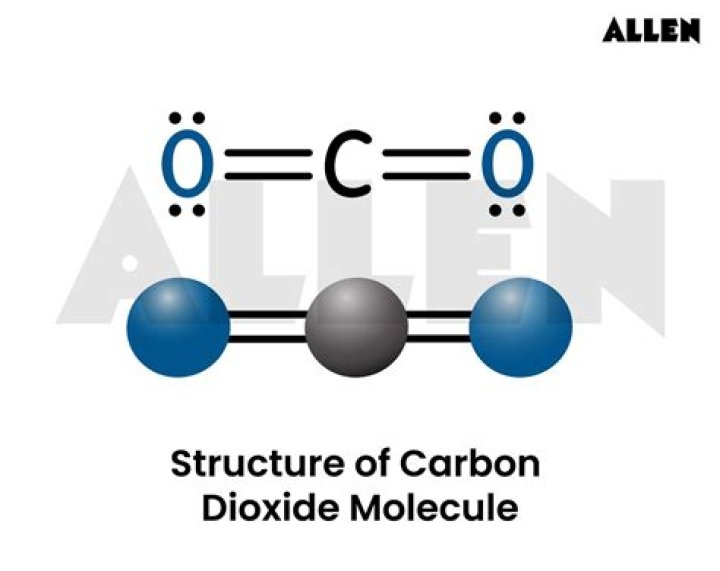
The molecular geometry of carbon dioxide is linear. This shape arises due to the arrangement of electron pairs around the central carbon atom. According to VSEPR theory, the repulsion between the two double bonds formed by carbon and oxygen results in a linear configuration.
Factors Influencing CO2 Geometry
- Electron Pair Repulsion: The lone pairs and bonding pairs of electrons repel each other, leading to the most stable arrangement.
- Double Bonds: The presence of double bonds between carbon and oxygen contributes to the linear shape.
- Electronegativity: Oxygen's higher electronegativity affects the bond angles and overall geometry.
The linear shape of CO2 ensures that the molecule is symmetrical, contributing to its nonpolar nature.
Bond Angle in Carbon Dioxide
The bond angle in carbon dioxide is 180 degrees. This perfect linear arrangement is a result of the equal repulsion between the two double bonds. The linear shape and 180-degree bond angle make CO2 a highly stable molecule.
Significance of Bond Angle
The bond angle in CO2 is crucial in determining its polarity. Since the molecule is symmetrical, the dipole moments of the C=O bonds cancel each other out, resulting in a nonpolar molecule.
This nonpolar nature has significant implications for CO2's solubility, reactivity, and interactions with other substances.
Electronic Structure and Lewis Dot Diagram
To understand the molecular shape of carbon dioxide, it is essential to examine its electronic structure. The Lewis dot diagram of CO2 illustrates the arrangement of valence electrons around the atoms.
Steps to Draw the Lewis Structure
- Count the total number of valence electrons: Carbon contributes 4, and each oxygen contributes 6, resulting in 16 valence electrons.
- Place the carbon atom in the center and the oxygen atoms on either side.
- Form double bonds between carbon and each oxygen atom to satisfy the octet rule.
The Lewis structure confirms the linear arrangement of atoms in CO2, with each oxygen atom sharing two pairs of electrons with the carbon atom.
Hybridization in Carbon Dioxide
Hybridization refers to the mixing of atomic orbitals to form new hybrid orbitals for bonding. In carbon dioxide, the carbon atom undergoes sp hybridization.
Understanding sp Hybridization
- sp Hybrid Orbitals: The carbon atom uses two sp hybrid orbitals to form sigma bonds with the oxygen atoms.
- Pi Bonds: The remaining unhybridized p orbitals overlap to form two pi bonds with each oxygen atom.
This sp hybridization results in a linear geometry, as the two sp hybrid orbitals are arranged at 180 degrees to minimize repulsion.
Properties Influenced by Molecular Shape
The molecular shape of carbon dioxide significantly influences its properties. Here are some key properties affected by its linear geometry:
Key Properties
- Nonpolar Nature: The symmetrical arrangement of atoms cancels out the dipole moments, making CO2 nonpolar.
- Solubility: CO2 dissolves in water to form carbonic acid, a weak acid that plays a role in the carbon cycle.
- Reactivity: The linear shape affects how CO2 reacts with other molecules, such as in photosynthesis and combustion processes.
Understanding these properties is essential for applications in industries like beverage carbonation and fire extinguishers.
Real-World Applications of CO2
Carbon dioxide finds applications in various fields due to its unique molecular shape and properties. Here are some real-world examples:
Applications
- Carbonated Beverages: CO2 is used to carbonate soft drinks, giving them their fizz.
- Fire Extinguishers: CO2 is effective in extinguishing fires due to its ability to smother flames.
- Greenhouse Gas: CO2 contributes to the greenhouse effect, playing a role in global climate change.
These applications highlight the importance of understanding the molecular shape and properties of CO2.
Comparison with Other Molecules
To better understand the molecular shape of carbon dioxide, it is helpful to compare it with other molecules. For instance, water (H2O) has a bent molecular shape due to the presence of lone pairs on the oxygen atom.
Differences in Geometry
- CO2 vs. H2O: While CO2 is linear, H2O is bent, resulting in different polarities and properties.
- CO2 vs. SO2: Sulfur dioxide (SO2) has a bent shape due to the lone pair on the sulfur atom, making it polar.
These comparisons illustrate how molecular shape influences the behavior and properties of different compounds.
Conclusion: Importance of Understanding CO2 Molecular Shape
In conclusion, the molecular shape of carbon dioxide is linear, a result of its sp hybridization and electron pair repulsion. This shape determines many of its properties, including its nonpolar nature and solubility in water. Understanding the molecular geometry of CO2 is crucial for applications in various fields, from beverage carbonation to climate science.
We invite you to share your thoughts and questions in the comments section below. For more insights into chemistry and related topics, explore our other articles and resources. Together, let's deepen our understanding of the fascinating world of molecules and their shapes!