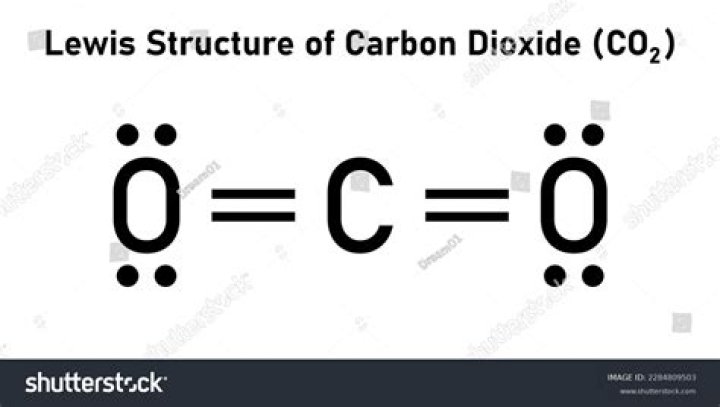
Carbon dioxide is a molecule that plays a critical role in our environment and biological processes. Understanding its structure is essential for comprehending how it interacts with other molecules and influences global climate patterns. This article dives deep into the structure of carbon dioxide, exploring its chemical properties, molecular geometry, and environmental implications.
As a greenhouse gas, carbon dioxide (CO2) significantly contributes to global warming and climate change. Its molecular structure determines how it absorbs and emits heat, making it a focal point for scientists and environmentalists worldwide.
This article aims to provide a detailed breakdown of the structure for carbon dioxide, offering insights into its molecular geometry, bond angles, and interactions with other substances. Whether you're a student, researcher, or simply curious about the science behind CO2, this guide will equip you with the necessary knowledge.
Introduction to Carbon Dioxide Structure
Carbon dioxide (CO2) is a colorless, odorless gas that consists of one carbon atom and two oxygen atoms. Its molecular structure is linear, with the carbon atom positioned symmetrically between the two oxygen atoms. This arrangement gives CO2 its unique properties and behavior in various environments.
The linear structure of carbon dioxide is crucial for understanding its role in the atmosphere. The molecule's symmetry and bond angles make it an effective greenhouse gas, capable of trapping heat and contributing to climate change. Scientists worldwide study the structure of CO2 to develop strategies for mitigating its effects on the planet.
In this section, we will explore the fundamental aspects of the structure for carbon dioxide, including its molecular geometry, bond characteristics, and interactions with other molecules. This foundational knowledge is vital for comprehending the broader implications of CO2 in both natural and industrial settings.
Molecular Geometry of Carbon Dioxide
The molecular geometry of carbon dioxide is linear, which means the molecule forms a straight line with the carbon atom at the center and the two oxygen atoms on either side. This linear arrangement arises due to the sp hybridization of the carbon atom, which creates two sigma bonds with the oxygen atoms.
In addition to the sigma bonds, carbon dioxide also features two pi bonds, resulting from the overlap of p orbitals. These pi bonds contribute to the molecule's stability and rigidity, reinforcing its linear shape.
Factors Influencing Molecular Geometry
- Electron Pair Repulsion: The absence of lone pairs on the central carbon atom ensures minimal repulsion, maintaining the linear geometry.
- Hybridization: The sp hybridization of the carbon atom contributes to the formation of strong sigma bonds, stabilizing the linear structure.
- Bond Length: The double bonds between carbon and oxygen result in shorter bond lengths, further reinforcing the molecule's rigidity.
Understanding Bond Angles in CO2
The bond angle in carbon dioxide is 180 degrees, a direct result of its linear molecular geometry. This perfect alignment minimizes electron pair repulsion and ensures optimal stability for the molecule.
Studies have shown that the linear structure and 180-degree bond angle of CO2 are critical for its role as a greenhouse gas. The symmetry of the molecule allows it to absorb infrared radiation efficiently, contributing to its warming effect on the Earth's atmosphere.
Importance of Bond Angles
- Stability: The 180-degree bond angle maximizes the distance between the oxygen atoms, reducing repulsion and increasing stability.
- Reactivity: The linear geometry affects how CO2 interacts with other molecules, influencing its reactivity in various chemical reactions.
- Optical Properties: The bond angle impacts the molecule's ability to absorb and emit light, which is essential for its role in the atmosphere.
Chemical Properties of Carbon Dioxide
Carbon dioxide exhibits several key chemical properties that make it unique. As a nonpolar molecule, CO2 does not dissolve easily in water, although it can react with water to form carbonic acid (H2CO3). This reaction is reversible and plays a crucial role in maintaining the Earth's carbon cycle.
Additionally, carbon dioxide is a weak acid, meaning it does not ionize completely in solution. This property makes it an important buffer in biological systems, helping to regulate pH levels in blood and other bodily fluids.
Key Chemical Properties
- Nonpolar Nature: The symmetric distribution of charge in CO2 makes it nonpolar, affecting its solubility and reactivity.
- Acidic Behavior: CO2 reacts with water to form carbonic acid, influencing pH levels in natural and biological systems.
- Reversible Reactions: The ability to form and break bonds with water allows CO2 to play a dynamic role in various chemical processes.
Environmental Impact of CO2
Carbon dioxide is one of the primary greenhouse gases contributing to global warming. Its ability to trap heat in the Earth's atmosphere has significant implications for climate change. Rising levels of CO2 are linked to increased temperatures, melting polar ice caps, and more frequent extreme weather events.
Efforts to reduce CO2 emissions are a global priority, with governments and organizations implementing policies to promote renewable energy and sustainable practices. Understanding the structure and behavior of carbon dioxide is essential for developing effective strategies to mitigate its environmental impact.
Global Efforts to Reduce CO2
- Renewable Energy: Transitioning to solar, wind, and hydroelectric power reduces reliance on fossil fuels, decreasing CO2 emissions.
- Carbon Capture Technology: Innovations in carbon capture and storage aim to remove CO2 from the atmosphere, addressing its contribution to climate change.
- Policy Implementation: International agreements like the Paris Accord set targets for reducing greenhouse gas emissions, encouraging global cooperation.
Biological Role of Carbon Dioxide
In biological systems, carbon dioxide plays a vital role in respiration and photosynthesis. During respiration, CO2 is produced as a byproduct of energy generation in cells. Conversely, during photosynthesis, plants absorb CO2 from the atmosphere and convert it into glucose, releasing oxygen in the process.
The balance between CO2 production and absorption is crucial for maintaining the Earth's carbon cycle. Disruptions to this cycle, such as deforestation or increased fossil fuel combustion, can have severe consequences for ecosystems and climate patterns.
Impact on Ecosystems
- Plant Growth: Elevated CO2 levels can enhance plant growth, but may also lead to nutrient imbalances and reduced biodiversity.
- Ocean Acidification: Increased CO2 absorption by oceans leads to acidification, threatening marine life and coral reefs.
- Climate Regulation: CO2's role in regulating global temperatures affects weather patterns and biodiversity.
Industrial Uses of Carbon Dioxide
Beyond its environmental and biological significance, carbon dioxide has numerous industrial applications. It is used in the production of carbonated beverages, fire extinguishers, and as a refrigerant in various industries. Additionally, CO2 is employed in enhanced oil recovery, where it is injected into oil reservoirs to increase extraction efficiency.
Advancements in technology continue to expand the industrial uses of carbon dioxide, promoting its recycling and reuse in innovative ways. These applications not only reduce waste but also contribute to more sustainable industrial practices.
Examples of Industrial Applications
- Food and Beverage Industry: CO2 is used to carbonate beverages and preserve food products.
- Fire Suppression: Its ability to displace oxygen makes CO2 an effective fire extinguishing agent.
- Enhanced Oil Recovery: CO2 injection improves oil extraction and reduces reliance on traditional methods.
Methods of Carbon Dioxide Production
Carbon dioxide is produced naturally through processes like respiration and volcanic activity. However, human activities such as burning fossil fuels, deforestation, and industrial processes significantly increase atmospheric CO2 levels. Understanding the sources of CO2 production is essential for addressing its environmental impact.
Efforts to reduce CO2 emissions focus on transitioning to cleaner energy sources and improving industrial processes to minimize waste and pollution. Innovations in carbon capture and utilization aim to transform CO2 into valuable resources, promoting a circular economy.
Primary Sources of CO2
- Fossil Fuel Combustion: Burning coal, oil, and natural gas releases large quantities of CO2 into the atmosphere.
- Deforestation: Clearing forests reduces the Earth's capacity to absorb CO2, exacerbating its buildup.
- Industrial Processes: Cement production and other industrial activities contribute significantly to CO2 emissions.
Reduction Methods for Carbon Dioxide
Reducing carbon dioxide emissions requires a multifaceted approach, combining technological innovation, policy implementation, and behavioral change. Strategies include transitioning to renewable energy sources, improving energy efficiency, and adopting sustainable practices in agriculture and industry.
Carbon capture and storage (CCS) technologies offer promising solutions for reducing atmospheric CO2 levels. These technologies capture CO2 emissions from industrial processes and store them underground, preventing their release into the atmosphere. Additionally, reforestation and afforestation projects aim to enhance the Earth's natural capacity to absorb CO2.
Innovative Solutions
- Carbon Capture Technology: Advances in CCS enable large-scale removal of CO2 from industrial emissions.
- Renewable Energy Adoption: Expanding the use of solar, wind, and hydroelectric power reduces reliance on fossil fuels.
- Reforestation Initiatives: Planting trees and restoring ecosystems enhances natural CO2 absorption.
Conclusion and Future Outlook
The structure for carbon dioxide plays a critical role in its behavior and interactions with the environment. Its linear geometry, bond angles, and chemical properties determine how it contributes to global warming and climate change. Understanding these aspects is essential for developing effective strategies to mitigate its environmental impact.
As we continue to explore innovative solutions for reducing CO2 emissions, it is crucial to prioritize sustainable practices and technologies. By transitioning to renewable energy, improving industrial processes, and enhancing natural carbon sinks, we can work towards a more sustainable future.
We invite you to share your thoughts and insights in the comments section below. Feel free to explore other articles on our site for more information on environmental science and sustainability. Together, we can make a difference in addressing the challenges posed by carbon dioxide and climate change.