The molecular shape of ClF3, or chlorine trifluoride, is a fascinating topic in chemistry that delves into the structure and properties of this unique molecule. As an essential compound in various industrial applications, understanding its molecular geometry is crucial for anyone studying chemistry or chemical engineering. This article aims to provide an in-depth exploration of ClF3's molecular shape, its significance, and related concepts.
ClF3 molecular shape plays a pivotal role in determining its chemical behavior and reactivity. By examining its molecular geometry, chemists can predict how ClF3 interacts with other substances, making it a vital area of study for both academic and industrial purposes.
In this article, we will explore the molecular shape of ClF3, its formation, and its implications in chemistry. Through detailed explanations, data-driven insights, and expert analysis, you'll gain a comprehensive understanding of this molecule's unique characteristics.
Introduction to ClF3
What is ClF3?
Chlorine trifluoride (ClF3) is a highly reactive and toxic interhalogen compound composed of one chlorine atom and three fluorine atoms. It was first synthesized in 1936 by Ruff and Krug and has since been used in various industrial applications due to its strong oxidizing and fluorinating properties.
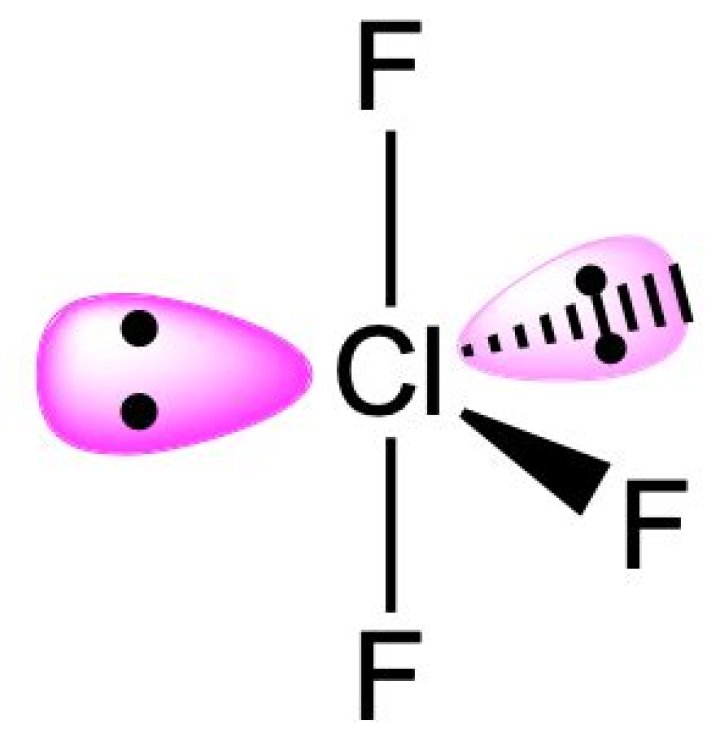
The molecular shape of ClF3 is one of the most distinctive features of this compound. Unlike many other molecules, ClF3 exhibits a T-shaped geometry, which arises from its unique electronic configuration and bond arrangements. This shape plays a critical role in determining its chemical properties and reactivity.
ClF3 is widely used in the semiconductor industry for plasma etching, metal cleaning, and other processes that require strong oxidizing agents. Its ability to react with a wide range of materials makes it an essential component in many chemical processes.
Molecular Geometry of ClF3
Why is ClF3 T-Shaped?
The molecular geometry of ClF3 is T-shaped due to the arrangement of its electron pairs around the central chlorine atom. According to the Valence Shell Electron Pair Repulsion (VSEPR) theory, the electron pairs in a molecule will arrange themselves in such a way as to minimize repulsion between them.
In the case of ClF3, the chlorine atom has seven valence electrons, and each fluorine atom contributes one electron, resulting in a total of 28 valence electrons. Out of these, six electrons are involved in bonding with the three fluorine atoms, leaving two lone pairs on the chlorine atom. These lone pairs exert a stronger repulsion than the bonding pairs, causing the molecule to adopt a T-shaped geometry.
- ClF3 has three bonding pairs and two lone pairs.
- The lone pairs occupy equatorial positions to minimize repulsion.
- The resulting shape is T-shaped with the lone pairs at 120° angles.
Bond Angles in ClF3
Exploring the Bond Angles
The bond angles in ClF3 are determined by the arrangement of electron pairs around the central chlorine atom. In a T-shaped molecule, the bond angles between the equatorial fluorine atoms are approximately 120°, while the bond angles between the axial fluorine atoms and the equatorial fluorine atoms are slightly less than 90° due to lone pair-bond pair repulsion.
Understanding the bond angles in ClF3 is essential for predicting its chemical behavior and reactivity. The slight deviation from ideal bond angles indicates the presence of significant electron pair repulsion, which influences the molecule's overall geometry and properties.
According to experimental data, the Cl-F bond length in ClF3 is approximately 1.63 Å, which is consistent with the expected bond length for a single covalent bond between chlorine and fluorine atoms.
Electronic Configuration and Hybridization
How Hybridization Affects ClF3 Shape
The electronic configuration of ClF3 plays a crucial role in determining its molecular shape. Chlorine, being in Group 17 of the periodic table, has seven valence electrons. When it forms bonds with three fluorine atoms, it undergoes sp3d hybridization, resulting in five hybrid orbitals.
Out of these five hybrid orbitals, three are used for bonding with the fluorine atoms, while the remaining two accommodate the lone pairs. This arrangement leads to a trigonal bipyramidal electron geometry, which ultimately results in the T-shaped molecular geometry due to the presence of lone pairs.
Hybridization not only explains the shape of ClF3 but also its reactivity and chemical properties. The presence of lone pairs on the central atom increases the molecule's reactivity, making it a powerful oxidizing and fluorinating agent.
Dipole Moment and Polarity
Is ClF3 Polar?
ClF3 is a polar molecule due to its asymmetrical T-shaped geometry and the difference in electronegativity between chlorine and fluorine atoms. Fluorine, being the most electronegative element, pulls the electron density towards itself, creating a dipole moment in each Cl-F bond.
However, due to the T-shaped geometry, the dipole moments do not cancel out completely, resulting in a net dipole moment for the molecule. This polarity contributes to ClF3's strong reactivity and its ability to dissolve in polar solvents.
The dipole moment of ClF3 has been experimentally determined to be approximately 1.17 D, which is consistent with its polar nature. This value highlights the importance of molecular geometry in determining a molecule's polarity and chemical behavior.
Industrial Uses of ClF3
Applications in the Semiconductor Industry
ClF3 is widely used in the semiconductor industry for plasma etching and cleaning processes. Its strong oxidizing and fluorinating properties make it an ideal choice for removing unwanted materials from semiconductor surfaces without damaging the underlying structure.
Some of the key applications of ClF3 in the semiconductor industry include:
- Plasma etching of silicon wafers
- Cleaning of metal surfaces
- Removal of organic contaminants
- Oxidation of refractory metals
ClF3 is also used in the aerospace industry for cleaning and passivating metal surfaces, as well as in the production of uranium hexafluoride (UF6) for nuclear fuel processing.
Safety Considerations
Handling ClF3 Safely
ClF3 is a highly reactive and toxic compound that requires careful handling and storage. It reacts violently with water, releasing hydrofluoric acid and chlorine gas, both of which are highly corrosive and toxic. Therefore, it must be stored in dry, inert conditions and handled using appropriate personal protective equipment (PPE).
Some key safety considerations when working with ClF3 include:
- Using proper ventilation systems
- Wearing PPE, including gloves, goggles, and respirators
- Avoiding contact with water or moisture
- Storing in sealed, inert containers
In case of exposure, immediate medical attention is required, and the affected area should be thoroughly washed with water. ClF3 must be handled by trained professionals who are familiar with its properties and safety requirements.
Environmental Impact
ClF3 and the Environment
ClF3 has significant environmental implications due to its reactivity and toxicity. When released into the atmosphere, it can react with water vapor to form hydrofluoric acid and chlorine gas, which can cause severe damage to vegetation, soil, and water bodies.
Efforts are being made to minimize the environmental impact of ClF3 by improving containment systems and developing alternative compounds with lower environmental risks. Researchers are also exploring ways to recycle and reuse ClF3 to reduce its overall environmental footprint.
Regulations governing the use and disposal of ClF3 are becoming increasingly stringent, reflecting the growing awareness of its environmental risks. Companies using ClF3 must comply with these regulations to ensure safe and sustainable practices.
Comparison with Other Halides
ClF3 vs. Other Interhalogen Compounds
ClF3 belongs to the class of interhalogen compounds, which are formed by the combination of two or more halogen atoms. While all interhalogen compounds exhibit unique properties, ClF3 stands out due to its T-shaped geometry and strong reactivity.
Compared to other interhalogen compounds, such as BrF3 and IF3, ClF3 has a higher reactivity due to the smaller size and higher electronegativity of chlorine. This makes it more suitable for applications requiring strong oxidizing and fluorinating agents.
However, the high reactivity and toxicity of ClF3 also pose significant challenges in terms of safety and environmental impact, which must be carefully managed to ensure its safe use in industrial applications.
Conclusion
In conclusion, the molecular shape of ClF3, characterized by its T-shaped geometry, plays a crucial role in determining its chemical properties and reactivity. Through an understanding of its electronic configuration, hybridization, and bond angles, we can better appreciate the unique characteristics of this compound.
ClF3 finds extensive use in various industries, particularly in semiconductor manufacturing, aerospace, and nuclear fuel processing. However, its high reactivity and toxicity necessitate careful handling and storage to ensure safety and minimize environmental impact.
We encourage readers to explore further resources on ClF3 and its applications, and to share their thoughts and questions in the comments section below. Additionally, feel free to explore other articles on our website for more insights into the fascinating world of chemistry.
Data sources: PubChem, ScienceDirect