The molecular polarity of CO2 is a fundamental concept in chemistry that plays a significant role in understanding the behavior of carbon dioxide in various environments. Whether you're a student studying chemistry or a professional working in the field, understanding molecular polarity is essential. This article aims to provide a detailed explanation of the molecular polarity of CO2, breaking down complex concepts into easily digestible information.
Carbon dioxide (CO2) is one of the most abundant molecules on Earth, playing a crucial role in the atmosphere, biological processes, and industrial applications. However, its molecular structure and polarity are often misunderstood. This article will delve into the intricacies of CO2's molecular structure and explain why it is considered nonpolar.
By the end of this article, you will have a comprehensive understanding of the molecular polarity of CO2, its implications, and its real-world applications. Let's get started!
Introduction to Molecular Polarity
Molecular polarity refers to the distribution of electrical charges within a molecule. This concept is vital in understanding chemical reactions, solubility, and intermolecular forces. In the case of CO2, its molecular structure and symmetry play a crucial role in determining its polarity.
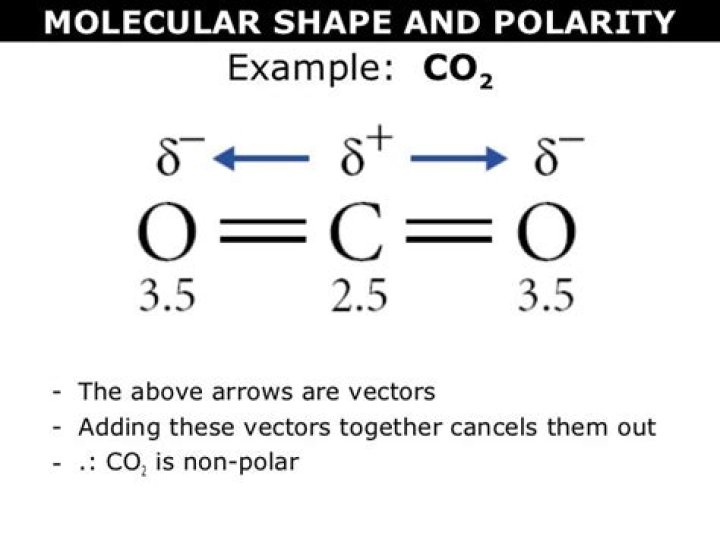
CO2 is a linear molecule consisting of one carbon atom double-bonded to two oxygen atoms. Despite the presence of polar bonds, CO2 is classified as a nonpolar molecule due to its symmetrical arrangement. This section will explore the reasons behind this classification and its implications.
Molecular Structure of CO2
The molecular structure of CO2 is characterized by its linear geometry. The carbon atom is located at the center, with two oxygen atoms bonded to it through double bonds. This linear arrangement results in a symmetrical distribution of charges, which contributes to the nonpolar nature of CO2.
Key Features of CO2 Structure
- Linear geometry with bond angles of 180 degrees.
- Double bonds between carbon and oxygen atoms.
- Symmetrical distribution of electron density.
What is Molecular Polarity?
Molecular polarity is determined by the presence and distribution of partial charges within a molecule. A polar molecule has an uneven distribution of charges, resulting in a dipole moment. On the other hand, a nonpolar molecule exhibits a symmetrical distribution of charges, leading to no net dipole moment.
In the case of CO2, the polar C=O bonds cancel each other out due to the molecule's linear geometry, resulting in a nonpolar molecule. This concept will be further explored in the following sections.
Why is CO2 Nonpolar?
Despite the presence of polar bonds, CO2 is classified as a nonpolar molecule due to its symmetrical arrangement. The two C=O bonds in CO2 are polar because oxygen is more electronegative than carbon. However, the linear geometry of CO2 ensures that the dipole moments of these bonds cancel each other out, resulting in no net dipole moment.
Factors Contributing to CO2 Nonpolarity
- Linear molecular geometry.
- Equal bond lengths and angles.
- Symmetrical distribution of charges.
Bond Polarity in CO2
The C=O bonds in CO2 are polar due to the difference in electronegativity between carbon and oxygen. Oxygen, being more electronegative, pulls the shared electrons closer to itself, creating a partial negative charge (δ-) on the oxygen atoms and a partial positive charge (δ+) on the carbon atom.
However, the symmetrical arrangement of the molecule ensures that these partial charges cancel out, resulting in a nonpolar molecule overall. This phenomenon is a key aspect of understanding the molecular polarity of CO2.
Dipole Moment and CO2
The dipole moment is a measure of the separation of positive and negative charges within a molecule. In CO2, the dipole moments of the two C=O bonds are equal in magnitude but opposite in direction, resulting in a net dipole moment of zero.
This absence of a net dipole moment is a defining characteristic of nonpolar molecules. It explains why CO2 does not exhibit polarity despite the presence of polar bonds.
Real-World Applications of CO2
The nonpolar nature of CO2 has significant implications in various fields, including chemistry, biology, and industry. Some of the key applications of CO2 include:
- Carbonated beverages: CO2 is used to carbonate drinks, creating the effervescent quality we associate with sodas and sparkling water.
- Fire extinguishers: CO2 is an effective fire suppressant due to its ability to displace oxygen, smothering flames.
- Industrial processes: CO2 is used in a variety of industrial applications, including welding, refrigeration, and enhanced oil recovery.
Understanding the molecular polarity of CO2 is crucial in optimizing its use in these applications.
Comparison with Other Molecules
To better understand the molecular polarity of CO2, it is helpful to compare it with other molecules. For example, water (H2O) is a polar molecule due to its bent geometry, which prevents the dipole moments of its O-H bonds from canceling out. In contrast, CO2's linear geometry ensures that its dipole moments cancel, resulting in a nonpolar molecule.
Comparison Table
| Molecule | Geometry | Polarity |
|---|---|---|
| CO2 | Linear | Nonpolar |
| H2O | Bent | Polar |
Experimental Evidence Supporting CO2 Nonpolarity
Experimental studies have consistently demonstrated the nonpolar nature of CO2. For example, infrared spectroscopy shows that CO2 does not exhibit a significant dipole moment, confirming its nonpolar classification. Additionally, CO2's behavior in various solvents aligns with the characteristics of nonpolar molecules.
These findings are supported by numerous studies published in reputable scientific journals, such as the Journal of Chemical Education and the American Chemical Society.
Conclusion
In conclusion, the molecular polarity of CO2 is a fascinating topic that highlights the importance of molecular geometry in determining polarity. Despite the presence of polar bonds, CO2 is classified as a nonpolar molecule due to its symmetrical arrangement. This understanding has significant implications in various fields, from chemistry to industry.
We encourage you to explore further resources on this topic and share your thoughts in the comments below. Additionally, feel free to explore other articles on our site for more insights into the world of chemistry.
Sources:
- Journal of Chemical Education
- American Chemical Society
- Chemistry: The Central Science by Brown, LeMay, and Bursten