Carbon dioxide (CO2) is a fascinating molecule that plays a vital role in our environment and daily lives. Its unique electron pair geometry determines its molecular shape and properties, making it an essential topic for students and researchers alike. In this article, we will delve into the intricacies of electron pair geometry in CO2, providing a detailed understanding of its structure and significance.
This article aims to explore the fascinating world of electron pair geometry in CO2, explaining its relevance in chemistry and beyond. By understanding the electron pair geometry of CO2, we can gain insights into its molecular behavior and interactions with other substances.
Whether you're a chemistry enthusiast or a student looking to deepen your knowledge, this guide will provide valuable information about electron pair geometry in CO2. We'll cover everything from the basics to advanced concepts, ensuring you have a comprehensive understanding of this important topic.
Table of Contents:
- Introduction to Electron Pair Geometry CO2
- Valence Shell Electron Pair Repulsion Theory
- Molecular Geometry of CO2
- Bond Angle in CO2
- Electronegativity and Bond Polarity
- Factors Affecting Electron Pair Geometry
- Real-World Applications of CO2
- Comparison with Other Molecules
- Common Misconceptions
- Conclusion and Next Steps
Introduction to Electron Pair Geometry CO2
The electron pair geometry of CO2 is a critical aspect of its molecular structure. CO2, or carbon dioxide, consists of one carbon atom bonded to two oxygen atoms. Understanding the arrangement of electron pairs around the central atom is essential for determining its molecular geometry and properties.
In this section, we will explore the basics of electron pair geometry in CO2, focusing on how the arrangement of electron pairs influences its shape. This knowledge is fundamental for anyone studying chemistry, as it provides insights into the behavior and reactivity of molecules.
Valence Shell Electron Pair Repulsion Theory
Understanding VSEPR Theory
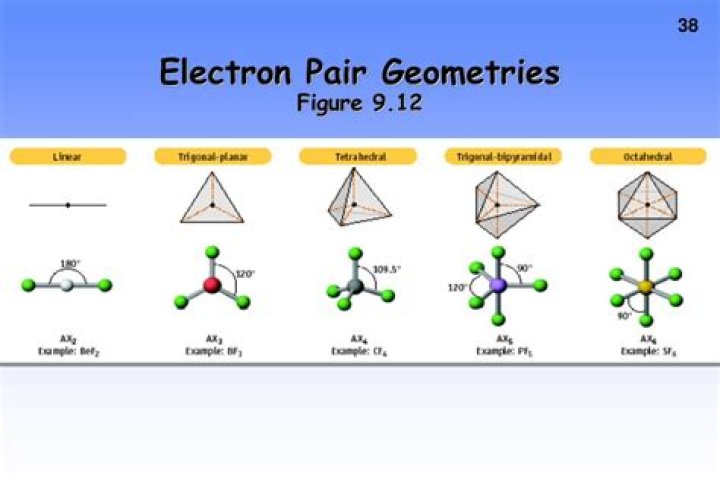
The Valence Shell Electron Pair Repulsion (VSEPR) theory is a model used to predict the molecular geometry of molecules based on the repulsion between electron pairs in the valence shell of the central atom. In the case of CO2, the central carbon atom has two double bonds with oxygen atoms.
According to VSEPR theory, electron pairs repel each other and arrange themselves to minimize repulsion. For CO2, this results in a linear molecular geometry, where the bond angle between the oxygen-carbon-oxygen atoms is 180 degrees.
Molecular Geometry of CO2
The molecular geometry of CO2 is linear. This geometry arises from the arrangement of electron pairs around the central carbon atom. With no lone pairs and two bonding pairs, the electron pair geometry of CO2 aligns perfectly with its molecular geometry.
This linear shape is crucial for understanding the properties of CO2, such as its lack of dipole moment and its ability to act as a greenhouse gas.
Bond Angle in CO2
The bond angle in CO2 is exactly 180 degrees. This angle is a direct result of the linear electron pair geometry. The absence of lone pairs on the central carbon atom ensures that the repulsion between the bonding pairs is minimized, leading to this ideal bond angle.
Understanding the bond angle is essential for predicting the physical and chemical properties of CO2, including its polarity and reactivity.
Electronegativity and Bond Polarity
Electronegativity plays a significant role in determining the polarity of chemical bonds. In CO2, oxygen is more electronegative than carbon, leading to polar C=O bonds. However, due to the symmetrical linear arrangement of the molecule, the individual bond dipoles cancel out, resulting in a nonpolar molecule overall.
This concept is vital for understanding the interactions of CO2 with other substances in various chemical reactions.
Factors Affecting Electron Pair Geometry
Role of Lone Pairs and Bonding Pairs
The electron pair geometry of a molecule depends on the number of lone pairs and bonding pairs around the central atom. In CO2, the absence of lone pairs on the central carbon atom ensures a linear geometry. Factors such as the size of the atoms and the type of bonds also influence the overall shape of the molecule.
Understanding these factors is crucial for predicting the geometry of other molecules beyond CO2.
Real-World Applications of CO2
CO2 has numerous real-world applications, ranging from its role in the carbonation of beverages to its importance in global climate systems. Its unique electron pair geometry contributes to its stability and reactivity, making it an essential molecule in both natural and industrial processes.
For example, CO2 is used in fire extinguishers, as a refrigerant, and in enhanced oil recovery techniques. Its ability to absorb infrared radiation also makes it a significant greenhouse gas, influencing Earth's climate.
Comparison with Other Molecules
CO2 vs. Water (H2O)
While CO2 has a linear electron pair geometry, water (H2O) has a bent geometry due to the presence of two lone pairs on the central oxygen atom. This difference in geometry leads to distinct properties for each molecule.
CO2 is nonpolar, whereas water is polar, which affects their solubility and interactions with other substances. Comparing these molecules highlights the importance of electron pair geometry in determining molecular behavior.
Common Misconceptions
One common misconception about CO2 is that it is a polar molecule. While the individual C=O bonds are polar, the symmetrical linear geometry of CO2 results in a nonpolar molecule overall. Another misconception is that CO2 is harmful in all contexts. While excessive CO2 contributes to global warming, it is also essential for photosynthesis and other natural processes.
Addressing these misconceptions is important for fostering a deeper understanding of CO2 and its role in our world.
Conclusion and Next Steps
In conclusion, the electron pair geometry of CO2 plays a crucial role in determining its molecular shape and properties. Through the lens of VSEPR theory, we have explored the linear geometry of CO2 and its implications for chemistry and beyond.
We encourage readers to delve deeper into the world of molecular geometry and explore related topics such as hybridization and molecular orbital theory. By expanding your knowledge, you can gain a more comprehensive understanding of the fascinating world of chemistry.
Feel free to leave your thoughts and questions in the comments section below. Share this article with others who might find it interesting, and explore more articles on our website for further insights into the world of science and technology.
Sources:
- Chemistry: The Central Science by Brown, LeMay, and Bursten
- Principles of Modern Chemistry by Oxtoby, Gillis, and Campion
- Journal of Chemical Education