CLF3 Lewis structure is an essential topic in chemistry that provides insights into the molecular geometry and bonding properties of this compound. If you're studying chemistry, understanding the CLF3 Lewis structure is crucial to grasp how atoms interact within molecules. This article will delve into the intricacies of CLF3, its Lewis structure, and its implications in chemical reactions.
Chemistry is not just about memorizing formulas; it's about understanding the behavior of molecules and how they interact. The CLF3 Lewis structure serves as a foundation for learning more complex concepts in molecular chemistry. By exploring this topic, you can deepen your knowledge of molecular bonding and its applications in real-world scenarios.
In this article, we will explore the CLF3 Lewis structure in detail, breaking down its components and explaining its significance. Whether you're a student, educator, or simply someone curious about chemistry, this guide will provide valuable insights into the world of molecular structures. Let's dive in!
Introduction to CLF3
CLF3, or chlorine trifluoride, is a highly reactive chemical compound used in various industrial applications. Understanding its Lewis structure is vital for predicting its chemical behavior and reactivity. CLF3 consists of one chlorine atom and three fluorine atoms, forming a unique molecular structure.
Properties of CLF3
CLF3 is a colorless gas at room temperature and exhibits strong oxidizing properties. Its reactivity makes it useful in applications such as rocket propellants and etching agents in semiconductor manufacturing. However, due to its high reactivity, handling CLF3 requires extreme caution.
Importance of CLF3 Lewis Structure
The Lewis structure of CLF3 provides insights into its bonding characteristics and molecular geometry. By analyzing the structure, chemists can predict how CLF3 interacts with other substances and its role in chemical reactions.
Lewis Structure Definition
A Lewis structure is a diagram that represents the bonding between atoms in a molecule and the lone pairs of electrons. It is named after Gilbert N. Lewis, who introduced this concept in 1916. The Lewis structure helps visualize the arrangement of electrons in a molecule, making it easier to understand its chemical properties.
Key Components of Lewis Structures
- Bonding pairs: Electrons shared between atoms to form covalent bonds.
- Lone pairs: Electrons not involved in bonding, located around central atoms.
- Valence electrons: Electrons in the outermost shell of an atom, responsible for bonding.
Drawing CLF3 Lewis Structure
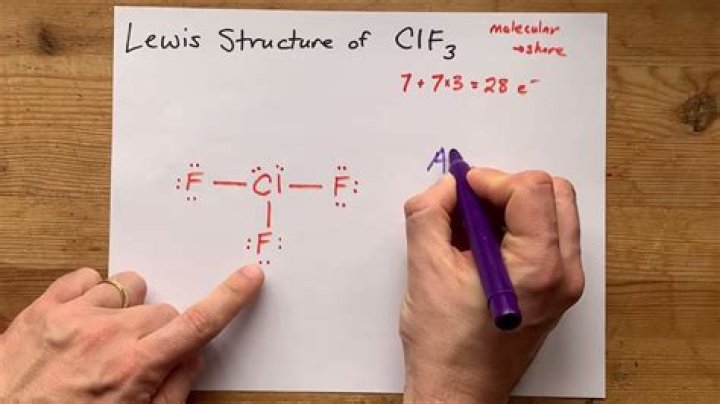
Drawing the CLF3 Lewis structure involves several steps. First, determine the total number of valence electrons in the molecule. Chlorine contributes seven valence electrons, and each fluorine atom contributes seven, resulting in a total of 28 valence electrons.
Steps to Draw the Structure
- Place the chlorine atom as the central atom, surrounded by three fluorine atoms.
- Connect each fluorine atom to chlorine with a single bond, using two electrons per bond.
- Distribute the remaining electrons as lone pairs around the atoms to satisfy the octet rule.
The resulting structure will show chlorine forming single bonds with three fluorine atoms, with two lone pairs of electrons remaining on the chlorine atom.
Molecular Geometry of CLF3
The molecular geometry of CLF3 is trigonal bipyramidal. This shape arises from the arrangement of electron pairs around the central chlorine atom. The three bonding pairs and two lone pairs arrange themselves to minimize repulsion, resulting in a trigonal bipyramidal geometry.
Factors Influencing Geometry
- Electron pair repulsion: Lone pairs exert more repulsion than bonding pairs, affecting the overall shape.
- Central atom: Chlorine's ability to expand its octet allows for the formation of this geometry.
Bond Angle in CLF3
In CLF3, the bond angle between the fluorine atoms is approximately 90 degrees and 120 degrees. These angles result from the trigonal bipyramidal geometry, where the lone pairs occupy axial positions, reducing repulsion with the bonding pairs.
Significance of Bond Angles
The bond angles in CLF3 play a crucial role in determining its reactivity and stability. The arrangement of atoms affects the molecule's polarity and its ability to participate in chemical reactions.
Electronic Configuration of CLF3
Understanding the electronic configuration of CLF3 involves analyzing the distribution of electrons in its orbitals. Chlorine, in its ground state, has the configuration [Ne] 3s² 3p⁵. When forming CLF3, chlorine expands its octet by utilizing its d-orbitals, allowing it to bond with three fluorine atoms.
Role of d-Orbitals
The involvement of d-orbitals in bonding is a unique feature of CLF3. This expansion of the octet enables the formation of stable molecules with more than eight electrons around the central atom.
Polarity of CLF3
CLF3 is a polar molecule due to the difference in electronegativity between chlorine and fluorine atoms. The fluorine atoms pull the shared electrons closer, creating a partial negative charge on the fluorine side and a partial positive charge on the chlorine side.
Factors Affecting Polarity
- Electronegativity difference: Fluorine's higher electronegativity contributes to the molecule's polarity.
- Molecular geometry: The asymmetrical arrangement of atoms enhances the polarity of CLF3.
Applications of CLF3
CLF3 finds applications in various industries due to its reactive nature. Some of its uses include:
- Rocket propellants: CLF3 serves as a powerful oxidizer in rocket fuel.
- Semiconductor manufacturing: It is used in the etching process for silicon wafers.
- Chemical synthesis: CLF3 acts as a fluorinating agent in organic synthesis.
Safety Considerations
Handling CLF3 requires strict safety measures due to its high reactivity and toxicity. Proper protective equipment and ventilation systems are essential when working with this compound.
Common Questions About CLF3
What is the Lewis structure of CLF3?
The Lewis structure of CLF3 consists of a central chlorine atom bonded to three fluorine atoms, with two lone pairs of electrons on the chlorine atom.
Why is CLF3 reactive?
CLF3 is reactive due to the presence of highly electronegative fluorine atoms and the availability of d-orbitals in chlorine, allowing it to form strong bonds with other substances.
Can CLF3 be stored safely?
Storing CLF3 safely requires specialized containers and conditions to prevent reactions with surrounding materials. It is generally stored in stainless steel containers under controlled environments.
Conclusion
In conclusion, understanding the CLF3 Lewis structure is essential for grasping the fundamental principles of molecular chemistry. By exploring its molecular geometry, bond angles, electronic configuration, and polarity, we gain insights into its chemical behavior and applications. CLF3's reactivity makes it valuable in various industries, but its handling requires caution.
We encourage you to leave your thoughts and questions in the comments section below. Share this article with others who might find it useful, and explore more content on our website to deepen your understanding of chemistry. Thank you for reading!