CLF3 electron geometry is an intriguing topic that delves into the molecular structure of chlorine trifluoride (CLF3). This compound, with its unique electron arrangement, plays a vital role in understanding chemical bonding and molecular geometry. By exploring the electron geometry of CLF3, we can gain insights into its physical and chemical properties, making it a fascinating subject for chemistry enthusiasts and professionals alike.
For those unfamiliar with the term, electron geometry refers to the three-dimensional arrangement of electron pairs around a central atom. In the case of CLF3, this arrangement significantly influences its behavior and reactivity. This article aims to provide a detailed explanation of CLF3 electron geometry, ensuring readers gain a comprehensive understanding of the topic.
Whether you're a student, researcher, or simply curious about the world of chemistry, this guide will serve as a valuable resource. We'll explore the intricacies of CLF3 electron geometry, its implications, and how it fits into broader chemical principles. Let's dive in!
Introduction to CLF3 Electron Geometry
Chlorine trifluoride (CLF3) is a compound composed of one chlorine atom and three fluorine atoms. Its electron geometry is determined by the arrangement of electron pairs around the central chlorine atom. Understanding this geometry is essential for predicting the compound's behavior in various chemical reactions.
Significance of Electron Geometry
Electron geometry is crucial in determining the shape of molecules. For CLF3, the geometry influences its polarity, reactivity, and other physical properties. By studying CLF3 electron geometry, chemists can better understand how this compound interacts with other substances.
Furthermore, electron geometry plays a role in the compound's stability and its ability to participate in chemical reactions. This knowledge is invaluable for applications in industries such as aerospace and electronics, where CLF3 is used as a cleaning agent and etchant.
Molecular Structure of CLF3
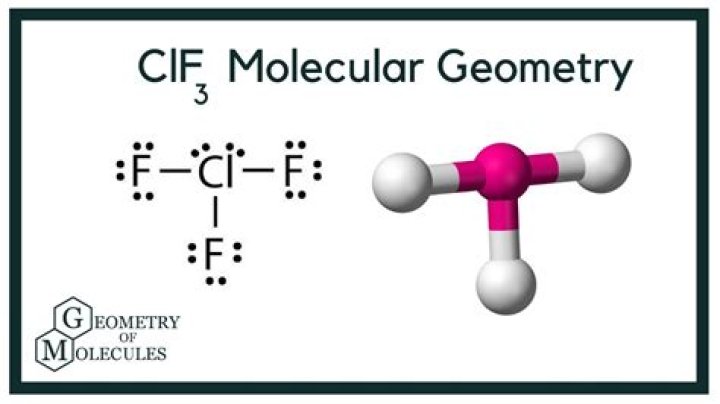
The molecular structure of CLF3 is defined by its electron geometry. In this compound, the central chlorine atom is bonded to three fluorine atoms and has two lone pairs of electrons. This arrangement results in a T-shaped molecular geometry.
Factors Affecting Molecular Structure
Several factors contribute to the molecular structure of CLF3, including:
- Electron repulsion between bonding pairs and lone pairs
- Number of electron domains around the central atom
- Hybridization of the central atom
These factors collectively determine the overall shape and properties of the molecule, making CLF3 a unique and interesting subject of study.
Applying VSEPR Theory to CLF3
The Valence Shell Electron Pair Repulsion (VSEPR) theory is a model used to predict the geometry of molecules based on the repulsion between electron pairs. Applying VSEPR theory to CLF3 reveals its T-shaped geometry, which arises from the repulsion between the five electron domains around the central chlorine atom.
Steps in Applying VSEPR Theory
To apply VSEPR theory to CLF3, follow these steps:
- Determine the Lewis structure of the molecule
- Identify the number of electron domains around the central atom
- Predict the electron geometry based on the number of domains
- Determine the molecular geometry by considering lone pairs
Through this process, we can accurately predict the geometry of CLF3 and understand its unique properties.
Bond Angles in CLF3
The bond angles in CLF3 are influenced by the repulsion between electron pairs. In a T-shaped geometry, the bond angles between the fluorine atoms are approximately 90 degrees and 180 degrees. These angles are slightly distorted due to the presence of lone pairs, which exert greater repulsion than bonding pairs.
Factors Affecting Bond Angles
Several factors affect the bond angles in CLF3, including:
- Electron pair repulsion
- Hybridization of the central atom
- Size and electronegativity of the surrounding atoms
Understanding these factors is essential for accurately predicting the geometry and properties of CLF3.
Electron Pair Arrangement in CLF3
The electron pair arrangement in CLF3 is determined by the VSEPR theory. The central chlorine atom has five electron domains: three bonding pairs and two lone pairs. This arrangement results in a trigonal bipyramidal electron geometry, which is distorted into a T-shaped molecular geometry due to the lone pairs.
Importance of Electron Pair Arrangement
The arrangement of electron pairs significantly influences the properties of CLF3. It affects the compound's polarity, reactivity, and overall stability. By understanding the electron pair arrangement, chemists can better predict how CLF3 will behave in various chemical environments.
Properties of CLF3
CLF3 exhibits several unique properties that make it valuable in various applications. Some of these properties include:
- Polarity: CLF3 is a polar molecule due to its asymmetrical shape and the difference in electronegativity between chlorine and fluorine.
- Reactivity: CLF3 is highly reactive and can react violently with water and other substances.
- Toxicity: The compound is toxic and requires careful handling in industrial and laboratory settings.
These properties make CLF3 a powerful yet dangerous substance, requiring proper safety measures during its use.
Applications of CLF3
Despite its reactivity and toxicity, CLF3 finds applications in several industries. Some of its key uses include:
- Aerospace: CLF3 is used as a cleaning agent for rocket engines and other high-temperature components.
- Electronics: The compound serves as an etchant in the semiconductor industry for cleaning and etching silicon wafers.
- Research: CLF3 is used in research labs to study chemical reactions and molecular structures.
Its unique properties make CLF3 indispensable in these fields, despite the challenges associated with its handling.
Comparison with Other Compounds
Comparing CLF3 with other compounds provides valuable insights into its unique characteristics. For instance, while both CLF3 and BF3 contain three fluorine atoms, their electron geometries differ significantly due to the presence of lone pairs in CLF3. This difference results in distinct properties and applications for each compound.
Key Differences
Some key differences between CLF3 and other compounds include:
- Molecular geometry
- Polarity
- Reactivity
Understanding these differences is essential for selecting the appropriate compound for specific applications.
Common Questions about CLF3 Electron Geometry
Many questions arise when studying CLF3 electron geometry. Below are some frequently asked questions and their answers:
What is the electron geometry of CLF3?
The electron geometry of CLF3 is trigonal bipyramidal, while its molecular geometry is T-shaped due to the presence of lone pairs.
Why is CLF3 highly reactive?
CLF3 is highly reactive because of its strong oxidizing and fluorinating properties, which allow it to react with a wide range of substances.
Is CLF3 polar or nonpolar?
CLF3 is a polar molecule due to its asymmetrical shape and the difference in electronegativity between chlorine and fluorine.
Conclusion and Call to Action
In conclusion, CLF3 electron geometry is a fascinating subject that offers valuable insights into the world of chemistry. By understanding the molecular structure, properties, and applications of CLF3, we can appreciate its significance in various industries and research fields. However, its reactivity and toxicity require careful handling to ensure safety.
We encourage readers to leave comments, share this article, or explore other articles on our site to deepen their knowledge of chemistry. Together, let's continue to explore the wonders of the chemical world!
For further reading, consider consulting reputable sources such as the Journal of Chemical Education and the American Chemical Society for additional insights into CLF3 and related compounds.