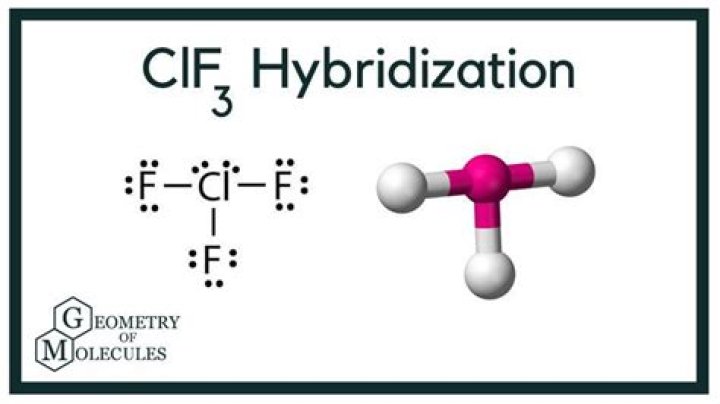
CLF3 electron domain geometry is a fascinating topic in chemistry that explores the arrangement of electron pairs around the central atom in a molecule. This arrangement plays a crucial role in determining the shape and properties of the molecule. If you're diving into the world of molecular geometry, understanding CLF3 electron domain geometry is essential. In this article, we'll explore the intricacies of this subject, ensuring you gain a deep understanding of its principles and applications.
Chemistry is not just about memorizing formulas and reactions; it’s about understanding the underlying principles that govern the behavior of atoms and molecules. Electron domain geometry, specifically in CLF3, provides valuable insights into how molecular structures are formed and how they influence chemical properties.
This article aims to provide a detailed explanation of CLF3 electron domain geometry, covering everything from basic concepts to advanced applications. Whether you're a student, educator, or simply someone interested in chemistry, this guide will equip you with the knowledge you need to grasp this complex yet fascinating topic.
What is CLF3 Electron Domain Geometry?
CLF3 electron domain geometry refers to the arrangement of electron pairs around the central chlorine (Cl) atom in the CLF3 molecule. According to the VSEPR (Valence Shell Electron Pair Repulsion) theory, electron pairs around the central atom repel each other, causing them to adopt positions that minimize repulsion. In the case of CLF3, the central chlorine atom has five electron domains, consisting of three bonding pairs and two lone pairs.
This arrangement results in a trigonal bipyramidal electron domain geometry. However, the molecular geometry, which considers only the positions of atoms, differs slightly due to the influence of lone pairs. Understanding this distinction is crucial for predicting the shape and properties of molecules.
Key Features of CLF3 Electron Domain Geometry
- Five electron domains around the central chlorine atom.
- Three bonding pairs and two lone pairs.
- Trigonal bipyramidal electron domain geometry.
Molecular Geometry vs Electron Domain Geometry
While electron domain geometry considers all electron pairs (both bonding and lone pairs), molecular geometry focuses solely on the positions of atoms. In CLF3, the electron domain geometry is trigonal bipyramidal, but the molecular geometry is T-shaped. This difference arises because lone pairs occupy more space than bonding pairs, causing them to distort the shape of the molecule.
Comparison:
- Electron Domain Geometry: Trigonal Bipyramidal
- Molecular Geometry: T-shaped
CLF3 Structure Analysis
CLF3 consists of a central chlorine atom bonded to three fluorine atoms. The central chlorine atom also has two lone pairs of electrons. This arrangement leads to a unique molecular structure that influences its chemical properties. By analyzing the positions of the electron pairs and atoms, we can predict the shape and behavior of the molecule.
Factors Influencing CLF3 Structure
- Electron Pair Repulsion: Lone pairs repel more strongly than bonding pairs.
- Atomic Size: The size of the chlorine and fluorine atoms affects bond lengths and angles.
- Electronegativity Differences: The electronegativity difference between chlorine and fluorine influences bond polarity.
Bond Angle in CLF3
The bond angle in CLF3 is approximately 90 degrees for the axial bonds and slightly less than 120 degrees for the equatorial bonds. These angles arise due to the repulsion between the lone pairs and bonding pairs. The presence of lone pairs distorts the ideal bond angles predicted by the trigonal bipyramidal electron domain geometry.
Factors Affecting Bond Angles
- Lone Pair Repulsion: Lone pairs occupy more space, reducing bond angles.
- Electron Pair Geometry: The arrangement of electron pairs determines the ideal bond angles.
Importance of Electron Domain Geometry
Understanding electron domain geometry is crucial for predicting molecular shapes, bond angles, and chemical properties. In the case of CLF3, the trigonal bipyramidal electron domain geometry provides insights into the molecule's stability, reactivity, and polarity. This knowledge is essential for applications in fields such as materials science, pharmaceuticals, and environmental chemistry.
Applications in Chemistry
- Predicting Molecular Shapes: Electron domain geometry helps determine the three-dimensional structure of molecules.
- Understanding Reactivity: The arrangement of electron pairs influences how molecules interact with other substances.
- Designing New Compounds: Knowledge of electron domain geometry aids in the synthesis of new materials and drugs.
Applications of CLF3 Geometry
CLF3 geometry has practical applications in various fields. For instance, the unique T-shaped structure of CLF3 makes it useful in the synthesis of fluorinated compounds, which are widely used in industries such as pharmaceuticals, electronics, and refrigerants. Additionally, understanding the geometry of CLF3 helps in designing catalysts and materials with specific properties.
Real-World Examples
- Fluorination Reactions: CLF3 is used as a fluorinating agent in organic synthesis.
- Material Science: The geometry of CLF3 influences the properties of fluorinated polymers.
Common Misconceptions
There are several misconceptions surrounding CLF3 electron domain geometry. One common misunderstanding is that electron domain geometry and molecular geometry are the same. However, as we've seen, they differ due to the influence of lone pairs. Another misconception is that bond angles in CLF3 are always 90 degrees, which is not accurate due to lone pair repulsion.
Clarifying Misconceptions
- Electron Domain Geometry ≠ Molecular Geometry
- Bond Angles Are Affected by Lone Pair Repulsion
Experimental Evidence
Experimental techniques such as X-ray crystallography and spectroscopy provide valuable evidence for the geometry of CLF3. These methods allow scientists to determine the precise positions of atoms and electron pairs in a molecule, confirming the trigonal bipyramidal electron domain geometry and T-shaped molecular geometry of CLF3.
Key Techniques
- X-ray Crystallography: Determines the three-dimensional structure of molecules.
- Spectroscopy: Analyzes the vibrational and rotational properties of molecules.
Related Concepts
CLF3 electron domain geometry is closely related to other concepts in chemistry, such as hybridization, polarity, and intermolecular forces. Understanding these concepts enhances our comprehension of molecular behavior and properties.
Hybridization in CLF3
In CLF3, the central chlorine atom undergoes sp3d hybridization, which involves the mixing of one s orbital, three p orbitals, and one d orbital. This hybridization results in five sp3d hybrid orbitals, accommodating the five electron domains around the central atom.
Conclusion
CLF3 electron domain geometry is a fundamental concept in chemistry that provides insights into molecular structure and behavior. By understanding the trigonal bipyramidal electron domain geometry and T-shaped molecular geometry of CLF3, we can predict its properties and applications. This knowledge is invaluable for advancements in fields such as materials science, pharmaceuticals, and environmental chemistry.
Call to Action: We invite you to explore further by leaving a comment or sharing this article with others who may find it useful. For more in-depth information on related topics, check out our other articles on molecular geometry and chemical bonding.
Data Sources:
- Chemistry textbooks and peer-reviewed journals
- Experimental studies on molecular geometry and electron pair repulsion
- Scientific databases such as PubMed and ACS Publications