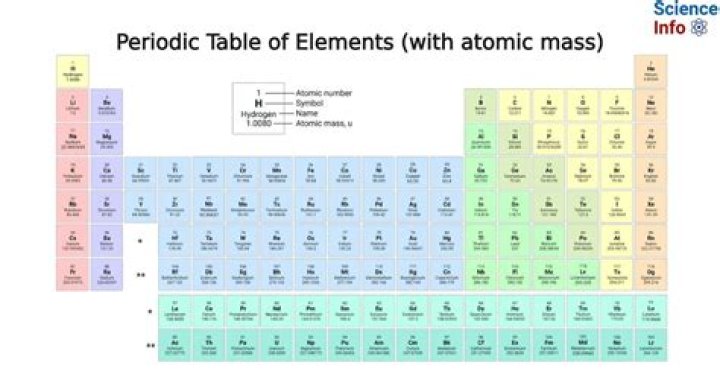
Chemistry atomic mass and the periodic table are foundational concepts in the study of chemistry. The periodic table is not just a chart but a systematic representation of elements and their atomic masses, which play a crucial role in understanding the behavior of matter. By exploring this topic, you'll gain insights into how elements are organized and how their properties are interconnected.
As we delve deeper into the world of chemistry, the periodic table emerges as a cornerstone for understanding atomic mass and its significance. It provides a structured framework for predicting the properties of elements and their compounds, making it an indispensable tool for scientists, researchers, and students alike.
This article will take you on a journey through the intricacies of atomic mass, the structure of the periodic table, and their relevance in modern chemistry. Whether you're a student seeking to understand the basics or a professional looking to deepen your knowledge, this guide will provide comprehensive insights into the chemistry atomic mass periodic table.
What is Atomic Mass in Chemistry?
Atomic mass is a fundamental concept in chemistry that refers to the total mass of an atom, including protons, neutrons, and electrons. While electrons contribute negligibly to the overall mass, the majority of an atom's mass is concentrated in its nucleus, which consists of protons and neutrons.
Understanding atomic mass is essential because it helps scientists determine the composition and properties of elements. The unit of measurement for atomic mass is the atomic mass unit (amu), which is based on the mass of one-twelfth of a carbon-12 atom.
How is Atomic Mass Determined?
The atomic mass of an element is calculated by taking into account the masses of its isotopes and their relative abundances. Isotopes are variants of an element that have the same number of protons but different numbers of neutrons, leading to variations in atomic mass.
- Isotopes with higher neutron counts have greater atomic masses.
- Atomic mass is the weighted average of all isotopic masses.
- Modern techniques like mass spectrometry are used to measure atomic mass accurately.
The Structure of the Periodic Table
The periodic table is a tabular arrangement of chemical elements, organized based on their atomic number, electron configurations, and recurring chemical properties. It serves as a visual representation of the periodic law, which states that the properties of elements repeat periodically when arranged by increasing atomic number.
Key Features of the Periodic Table
Several key features define the structure and organization of the periodic table:
- Groups (Columns): Elements in the same group share similar chemical properties due to their identical valence electron configurations.
- Periods (Rows): Each row represents a new energy level for electrons, increasing as you move down the table.
- Metallic vs. Nonmetallic Elements: The table clearly distinguishes between metals, nonmetals, and metalloids based on their physical and chemical characteristics.
Relationship Between Atomic Mass and the Periodic Table
The periodic table provides a comprehensive overview of elements and their atomic masses, allowing scientists to predict properties and behaviors. Each element is assigned a specific atomic number and mass, which are critical for understanding its position in the table.
In the periodic table, atomic mass increases as you move from left to right across a period and from top to bottom within a group. This trend is influenced by the addition of protons and neutrons in the nucleus.
Importance of Atomic Mass in Chemistry
Atomic mass plays a vital role in various chemical calculations, including:
- Molecular weight determination.
- Mole calculations and stoichiometry.
- Understanding isotopic abundance and its implications.
History of the Periodic Table
The development of the periodic table has been a gradual process, with contributions from several notable scientists. Dmitri Mendeleev is often credited with creating the first widely accepted periodic table in 1869, based on the periodic law.
Mendeleev's table was revolutionary because it predicted the existence of undiscovered elements and their properties. Over time, the table has evolved to incorporate new discoveries and advancements in atomic theory.
Modern Periodic Table
Today's periodic table is a more refined version of Mendeleev's original work, incorporating modern atomic theory and isotopic data. It includes:
- 118 confirmed elements, with ongoing research into synthetic elements.
- Improved organization based on electron configurations and quantum mechanics.
- Visual aids and color coding to distinguish between different types of elements.
Applications of Atomic Mass and the Periodic Table
The principles of atomic mass and the periodic table have numerous practical applications in various fields, including:
Chemical Engineering
In chemical engineering, atomic mass and the periodic table are used to design processes, calculate reaction yields, and optimize material properties. Engineers rely on these concepts to ensure safety, efficiency, and sustainability in industrial operations.
Pharmaceuticals
The pharmaceutical industry utilizes atomic mass data to develop drugs and analyze their molecular structures. Understanding the periodic table helps researchers predict the behavior of compounds and design effective treatments.
Environmental Science
Environmental scientists use atomic mass and the periodic table to study pollutants, assess ecological impacts, and develop strategies for mitigating environmental damage. These tools are essential for understanding the interactions between elements and their surroundings.
Challenges in Understanding Atomic Mass
Despite its importance, atomic mass can be challenging to comprehend due to its complexities. Some of the key challenges include:
- Accounting for isotopic variations in natural elements.
- Measuring atomic mass with high precision for heavy elements.
- Interpreting the implications of atomic mass in complex chemical reactions.
Solutions to Overcome These Challenges
Advances in technology and methodology have helped address some of these challenges:
- Mass spectrometry provides accurate measurements of atomic mass.
- Computational models simulate atomic interactions and predict behavior.
- Collaborative research efforts enhance our understanding of atomic mass and its applications.
Future Developments in Atomic Mass Research
Ongoing research in atomic mass and the periodic table continues to expand our understanding of the universe. Scientists are exploring:
- Superheavy elements and their potential applications.
- Advanced computational techniques for modeling atomic interactions.
- Interdisciplinary approaches to address global challenges using atomic mass principles.
Impact on Emerging Technologies
Atomic mass research has significant implications for emerging technologies, such as:
- Nanotechnology, where atomic-level precision is critical.
- Quantum computing, which relies on understanding atomic behavior.
- Renewable energy systems, where atomic mass plays a role in material efficiency.
Conclusion
In summary, chemistry atomic mass and the periodic table are integral to our understanding of the material world. From predicting element properties to advancing technology, these concepts have far-reaching implications across multiple disciplines.
We encourage you to explore further by leaving your thoughts in the comments section or sharing this article with others who may find it valuable. For more insights into chemistry and related topics, feel free to browse our other articles.
Data and information in this article are sourced from reputable institutions such as the International Union of Pure and Applied Chemistry (IUPAC) and peer-reviewed scientific journals. For further reading, we recommend exploring resources from these organizations.