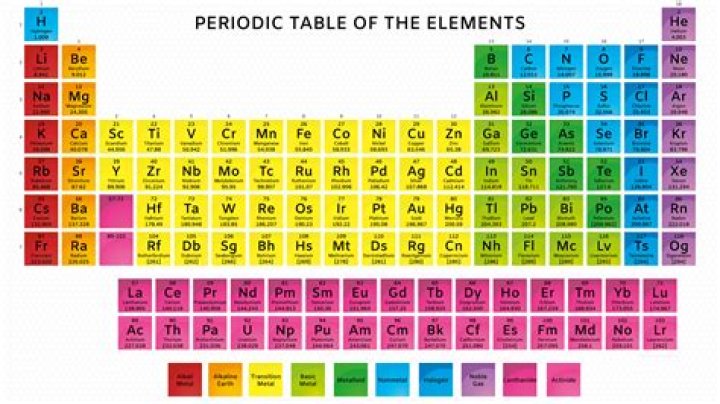
The periodic table for mass is an essential tool used in chemistry and physics to understand the atomic structure of elements. It provides crucial information about the atomic mass of each element, which is vital for various scientific calculations and applications. By studying the periodic table, we gain insights into the properties of elements and how they interact with one another.
The periodic table is not just a random arrangement of elements but a carefully organized chart that reflects the periodic trends and properties of all known elements. Understanding the concept of atomic mass is fundamental for anyone interested in science, as it forms the basis of many chemical reactions and physical phenomena.
This article delves deep into the periodic table for mass, exploring its structure, significance, and applications. Whether you're a student, educator, or science enthusiast, this guide will provide you with valuable insights into the world of atomic mass and its importance in modern science.
History of the Periodic Table
The periodic table has a rich history that dates back to the 19th century. Dmitri Mendeleev, a Russian chemist, is credited with creating the first periodic table in 1869. His groundbreaking work organized elements based on their atomic weights and chemical properties. Mendeleev's periodic table was revolutionary because it predicted the existence of undiscovered elements, which were later confirmed by scientific discoveries.
Over the years, the periodic table has evolved significantly. The modern periodic table is based on atomic number rather than atomic weight, providing a more accurate representation of the elements. This evolution has allowed scientists to better understand the periodic trends and relationships between elements, including their atomic mass.
Understanding Atomic Mass
Atomic mass refers to the total mass of an atom, which is primarily determined by the number of protons and neutrons in its nucleus. It is measured in atomic mass units (amu) and is a crucial parameter in chemistry and physics. The atomic mass of an element is usually listed below its symbol in the periodic table.
Variations in Atomic Mass
While the atomic mass of an element is generally consistent, variations can occur due to the presence of isotopes. Isotopes are atoms of the same element with different numbers of neutrons, resulting in variations in atomic mass. Understanding these variations is essential for accurate scientific calculations.
Periodic Trends in Mass
The periodic table exhibits several trends related to atomic mass. As you move from left to right across a period, the atomic mass generally increases due to the addition of protons and neutrons. Similarly, moving down a group, the atomic mass tends to increase because of the increasing number of electron shells.
These periodic trends are vital for predicting the behavior of elements in various chemical reactions. By understanding these trends, scientists can make informed decisions about the compatibility and reactivity of different elements.
Properties of Elements Based on Mass
The atomic mass of an element influences its physical and chemical properties. For example, elements with higher atomic masses tend to have higher densities and melting points. Additionally, atomic mass plays a significant role in determining the reactivity of elements, especially in chemical reactions involving ions and molecules.
Key Properties Influenced by Atomic Mass
- Density
- Melting and boiling points
- Reactivity
- Electron affinity
Isotopes and Their Role in Atomic Mass
Isotopes are atoms of the same element with different numbers of neutrons, leading to variations in atomic mass. These variations are reflected in the average atomic mass listed in the periodic table, which is a weighted average of all naturally occurring isotopes of an element.
Understanding isotopes is crucial in fields such as nuclear physics, where isotopes are used in various applications, including power generation and medical imaging. The study of isotopes also provides insights into the origins of elements and the processes that govern their formation.
Applications of Atomic Mass
The concept of atomic mass has numerous practical applications across various fields. Below are some of the most significant applications:
In Chemistry
In chemistry, atomic mass is used to calculate the molar mass of compounds, which is essential for stoichiometric calculations. It helps chemists determine the amounts of reactants and products in chemical reactions, ensuring precise and accurate results.
In Physics
Atomic mass plays a critical role in nuclear physics, where it is used to study the behavior of atomic nuclei and the energy released during nuclear reactions. This knowledge is applied in the development of nuclear power plants and atomic weapons.
In Medicine
In medicine, isotopes are used in diagnostic imaging and cancer treatment. Radioactive isotopes emit radiation that can be detected by imaging equipment, allowing doctors to visualize internal structures and diagnose diseases. Additionally, isotopes are used in radiation therapy to target and destroy cancer cells.
How to Calculate Atomic Mass
Calculating atomic mass involves determining the weighted average of the masses of all naturally occurring isotopes of an element. This calculation considers the relative abundance of each isotope, ensuring an accurate representation of the element's atomic mass.
Formula for Calculating Atomic Mass:
Atomic Mass = (Mass of Isotope 1 × Abundance of Isotope 1) + (Mass of Isotope 2 × Abundance of Isotope 2) + ...
Mass Spectrum and Its Relevance
A mass spectrum is a graphical representation of the masses of ions produced during a mass spectrometry analysis. It provides valuable information about the atomic and molecular composition of a sample, including the presence of isotopes and their relative abundances.
Mass spectrometry is widely used in fields such as chemistry, biology, and environmental science to analyze complex mixtures and identify unknown compounds. The data obtained from mass spectrometry can be used to calculate atomic masses and study the behavior of elements in various environments.
Future Developments in the Periodic Table
As scientific research continues to advance, the periodic table is likely to undergo further developments. The discovery of new elements and isotopes will expand our understanding of atomic mass and its implications. Additionally, advancements in technology may lead to new methods for studying and analyzing the properties of elements, providing even greater insights into their behavior.
Researchers are also exploring the potential for creating synthetic elements with unique properties, which could revolutionize various industries. These developments highlight the importance of the periodic table as a dynamic and evolving tool in the field of science.
Conclusion
The periodic table for mass is an invaluable resource for anyone interested in science. By understanding the atomic mass of elements and their periodic trends, we gain insights into the fundamental building blocks of matter and their interactions. This knowledge has numerous applications in fields such as chemistry, physics, and medicine, driving innovation and progress.
We encourage readers to explore the periodic table further and apply its principles in their studies and research. Feel free to leave comments or questions below, and don't forget to share this article with others who may find it useful. For more information on related topics, check out our other articles on this website.