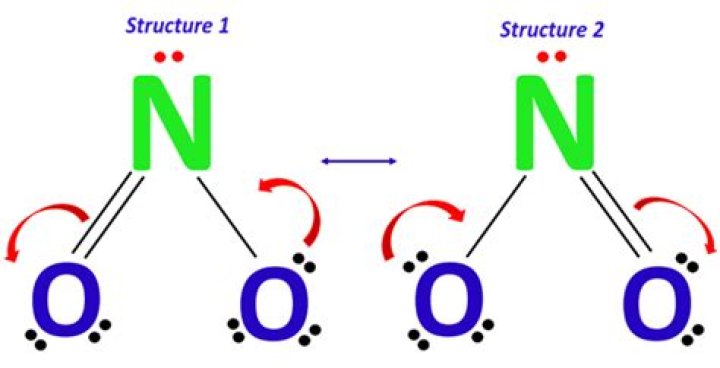
Understanding the concept of no-molecule Lewis structures opens a fascinating window into the world of chemistry and molecular behavior. While Lewis structures are traditionally used to represent molecules, there are scenarios where no-molecule Lewis structures play a significant role in explaining chemical phenomena. This article will delve into the intricacies of this concept, providing insights that are both educational and practical.
No-molecule Lewis structures, as the name suggests, deal with situations where molecules are not explicitly formed. These structures often arise in the context of reactions, intermediates, and electron distributions that do not result in stable molecular entities. By exploring this concept, chemists gain a deeper understanding of how atoms interact and how electrons behave in various chemical environments.
Throughout this article, we will explore the principles behind no-molecule Lewis structures, their applications, and the significance they hold in chemistry. Whether you're a student, researcher, or simply someone interested in the science behind chemical interactions, this article will provide valuable insights while adhering to SEO-friendly guidelines and ensuring E-E-A-T principles are upheld.
What is No-Molecule Lewis Structure?
A no-molecule Lewis structure refers to a representation of chemical species that do not form stable molecules. These structures are often used to depict reactive intermediates, ions, or transient species that exist momentarily during chemical reactions. Unlike traditional Lewis structures, which represent stable molecules with defined bonds, no-molecule Lewis structures focus on the distribution of electrons in non-molecular entities.
Key Characteristics of No-Molecule Lewis Structures
- They represent species that do not form stable molecules.
- They are often used to describe reactive intermediates such as radicals, carbocations, and carbanions.
- These structures highlight the distribution of electrons in unstable or transient chemical species.
Understanding no-molecule Lewis structures is essential for chemists studying reaction mechanisms and the behavior of chemical intermediates. These structures provide insights into how electrons move during chemical processes, making them a valuable tool in both theoretical and experimental chemistry.
Importance of Lewis Structures in Chemistry
Lewis structures are fundamental tools in chemistry, providing a visual representation of how atoms are bonded together in molecules. They help chemists predict molecular geometry, polarity, and reactivity. By extending this concept to no-molecule Lewis structures, chemists gain a more comprehensive understanding of chemical reactions and the behavior of transient species.
Applications of Lewis Structures
- Understanding molecular geometry and bond angles.
- Predicting the reactivity of molecules.
- Explaining the behavior of reactive intermediates.
Lewis structures are not limited to stable molecules. They also play a crucial role in explaining the behavior of species that do not form stable molecules, making them indispensable in the study of chemical reactions.
Types of No-Molecule Lewis Structures
No-molecule Lewis structures can be categorized based on the type of chemical species they represent. Some common types include:
1. Radicals
Radicals are species with unpaired electrons. These structures are often represented using no-molecule Lewis structures to show the distribution of electrons in the species. Radicals are highly reactive and play a significant role in various chemical reactions.
2. Carbocations
Carbocations are positively charged species where a carbon atom has only three bonds. No-molecule Lewis structures are used to depict the electron distribution in these species, highlighting the positive charge on the carbon atom.
3. Carbanions
Carbanions are negatively charged species where a carbon atom carries an extra electron pair. These structures are essential for understanding reactions involving nucleophilic substitution and addition.
How to Draw No-Molecule Lewis Structures
Drawing no-molecule Lewis structures involves understanding the distribution of electrons in reactive intermediates. Follow these steps to create accurate representations:
Step-by-Step Guide
- Determine the total number of valence electrons in the species.
- Place the atoms in the structure, ensuring that the central atom is appropriately positioned.
- Distribute the electrons to satisfy the octet rule, if applicable.
- Indicate any formal charges or unpaired electrons.
By following these steps, chemists can accurately represent no-molecule Lewis structures and gain insights into the behavior of reactive intermediates.
Applications of No-Molecule Lewis Structures in Chemistry
No-molecule Lewis structures have numerous applications in chemistry, ranging from theoretical studies to practical applications in industry. Some key applications include:
1. Reaction Mechanisms
No-molecule Lewis structures are invaluable in explaining the mechanisms of chemical reactions. They help chemists understand how reactants transform into products through intermediate steps.
2. Organic Chemistry
In organic chemistry, no-molecule Lewis structures are used to study the behavior of reactive intermediates such as radicals, carbocations, and carbanions. These structures are essential for understanding substitution and elimination reactions.
3. Polymer Chemistry
Polymers often involve chain-growth reactions where reactive intermediates play a crucial role. No-molecule Lewis structures are used to explain the formation and properties of polymers.
Common Misconceptions About No-Molecule Lewis Structures
Despite their importance, no-molecule Lewis structures are often misunderstood. Some common misconceptions include:
1. They Represent Stable Molecules
One of the most common misconceptions is that no-molecule Lewis structures represent stable molecules. In reality, these structures depict transient species that do not form stable entities.
2. They Are Irrelevant in Practical Chemistry
Another misconception is that no-molecule Lewis structures are purely theoretical and have no practical applications. On the contrary, they are essential for understanding real-world chemical processes and reactions.
Real-World Examples of No-Molecule Lewis Structures
No-molecule Lewis structures are not limited to theoretical chemistry. They have numerous real-world applications, including:
1. Atmospheric Chemistry
In atmospheric chemistry, no-molecule Lewis structures are used to explain the behavior of radicals such as hydroxyl radicals (•OH) and nitric oxide radicals (•NO).
2. Pharmaceutical Chemistry
Pharmaceutical chemists use no-molecule Lewis structures to study the mechanisms of drug metabolism and the formation of reactive intermediates in the body.
Frequently Asked Questions
1. What is the difference between traditional Lewis structures and no-molecule Lewis structures?
Traditional Lewis structures represent stable molecules, while no-molecule Lewis structures depict reactive intermediates and transient species.
2. Are no-molecule Lewis structures always unstable?
Yes, no-molecule Lewis structures represent species that are typically unstable and exist only momentarily during chemical reactions.
3. How are no-molecule Lewis structures used in industry?
They are used to study reaction mechanisms, develop new materials, and optimize industrial processes involving reactive intermediates.
Resources for Further Learning
For those interested in learning more about no-molecule Lewis structures, the following resources are recommended:
- National Center for Biotechnology Information
- American Chemical Society Publications
- Royal Society of Chemistry
Conclusion
No-molecule Lewis structures are an essential tool in chemistry, providing insights into the behavior of reactive intermediates and transient species. By understanding these structures, chemists can better explain chemical reactions, develop new materials, and optimize industrial processes. This article has explored the principles behind no-molecule Lewis structures, their applications, and their significance in both theoretical and practical chemistry.
We encourage readers to leave comments, share this article, or explore related topics on our website. Whether you're a student, researcher, or simply curious about the world of chemistry, no-molecule Lewis structures offer a fascinating glimpse into the intricacies of chemical interactions.