Carbon dioxide (CO2) is one of the most important molecules in our atmosphere, playing a crucial role in the Earth's ecosystem. Its molecular structure is fascinating and vital for understanding its properties and behavior. CO2 is not just a greenhouse gas but also a molecule with a unique linear structure that dictates its chemical and physical characteristics.
The molecular structure for CO2 has been studied extensively due to its environmental significance and industrial applications. Understanding the arrangement of atoms and bonds within this molecule helps scientists predict its reactivity, stability, and interactions with other substances. This knowledge is essential for addressing climate change and developing sustainable technologies.
In this article, we will delve into the molecular structure of CO2, exploring its geometry, bond angles, and properties. We'll also discuss its role in the environment and various applications. Whether you're a student, researcher, or simply curious about the science behind CO2, this article provides comprehensive insights into the molecular structure of carbon dioxide.
Introduction to CO2
Carbon dioxide (CO2) is a colorless and odorless gas that consists of one carbon atom and two oxygen atoms. It is a naturally occurring molecule found in the Earth's atmosphere and plays a critical role in the carbon cycle. CO2 is essential for plant photosynthesis and is also a byproduct of respiration in living organisms.
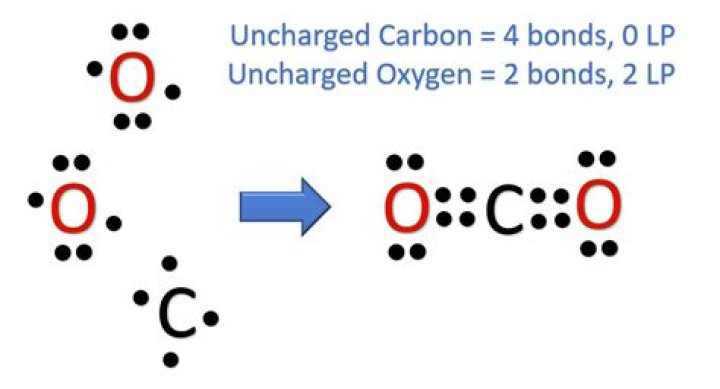
The molecular structure for CO2 is linear, with the carbon atom situated in the center and the two oxygen atoms bonded to it. This arrangement gives CO2 its unique properties, such as being nonpolar and highly stable. Understanding the structure of CO2 is crucial for predicting its behavior in various environments.
Molecular Geometry of CO2
Understanding Linear Geometry
The molecular geometry of CO2 is linear, meaning the atoms are arranged in a straight line. This geometry arises due to the sp hybridization of the carbon atom, which forms two double bonds with the oxygen atoms. The bond angle in CO2 is 180 degrees, making it a perfectly symmetrical molecule.
Here are the key features of CO2's molecular geometry:
- Linear shape with a bond angle of 180 degrees.
- Sp hybridization of the carbon atom.
- Two double bonds between carbon and oxygen atoms.
Bonding in CO2
Double Bonds and Electron Sharing
In CO2, the carbon atom forms two double bonds with the oxygen atoms. Each double bond consists of one sigma (σ) bond and one pi (π) bond. The sigma bond is formed by the head-on overlap of orbitals, while the pi bond is formed by the side-to-side overlap of p orbitals.
This strong double bonding contributes to the stability of CO2, making it a non-reactive molecule under normal conditions. However, under extreme conditions, such as high temperatures or pressures, CO2 can undergo chemical reactions.
Properties of CO2
Physical and Chemical Characteristics
CO2 exhibits several unique properties due to its molecular structure:
- Nonpolar: The symmetrical arrangement of atoms in CO2 makes it a nonpolar molecule.
- High Stability: The strong double bonds in CO2 contribute to its stability and low reactivity.
- Greenhouse Gas: CO2 traps heat in the Earth's atmosphere, contributing to the greenhouse effect.
These properties make CO2 an important molecule in both natural and industrial processes.
Environmental Impact of CO2
Role in Climate Change
CO2 is a major contributor to climate change due to its ability to trap heat in the Earth's atmosphere. Human activities, such as burning fossil fuels and deforestation, have significantly increased the concentration of CO2 in the atmosphere. This rise in CO2 levels leads to global warming, melting ice caps, and rising sea levels.
According to the Intergovernmental Panel on Climate Change (IPCC), the current concentration of CO2 in the atmosphere is over 400 parts per million (ppm), a level not seen in millions of years. Reducing CO2 emissions is critical for mitigating the impacts of climate change.
Industrial Applications of CO2
Uses in Various Industries
Despite its role as a greenhouse gas, CO2 has numerous industrial applications:
- Food and Beverage Industry: CO2 is used in carbonated beverages, refrigeration, and food preservation.
- Oil and Gas Industry: CO2 is employed in enhanced oil recovery techniques.
- Fire Suppression: CO2 is a common component in fire extinguishers.
These applications highlight the versatility of CO2 in various sectors, making it an indispensable molecule in modern industries.
Variations of CO2
Different Forms of Carbon Dioxide
CO2 exists in different forms depending on temperature and pressure:
- Gas: CO2 is a gas at standard temperature and pressure.
- Liquid: Under high pressure, CO2 can exist as a liquid.
- Solid (Dry Ice): At very low temperatures, CO2 solidifies into dry ice, which is widely used for cooling purposes.
These variations allow CO2 to be utilized in diverse applications, from cooling systems to medical treatments.
Scientific Research on CO2
Advancements in Understanding CO2
Scientific research on CO2 has advanced significantly over the years. Researchers are exploring new ways to capture and utilize CO2, such as carbon capture and storage (CCS) technologies. These innovations aim to reduce CO2 emissions and combat climate change.
Studies also focus on understanding the molecular structure of CO2 and its interactions with other substances. This knowledge is vital for developing new materials and technologies that can harness the potential of CO2 for sustainable development.
Frequently Asked Questions
Common Questions About CO2
Here are some frequently asked questions about CO2:
- What is the molecular formula of CO2? The molecular formula of CO2 is one carbon atom and two oxygen atoms.
- Why is CO2 a greenhouse gas? CO2 traps heat in the Earth's atmosphere, contributing to the greenhouse effect.
- How can we reduce CO2 emissions? Reducing CO2 emissions involves adopting renewable energy sources, improving energy efficiency, and implementing carbon capture technologies.
Conclusion
The molecular structure for CO2 is a fascinating subject that sheds light on its properties and behavior. From its linear geometry to its role in the environment, CO2 is a molecule of great importance. Understanding its structure and properties is essential for addressing global challenges such as climate change and developing sustainable technologies.
We encourage readers to share their thoughts and questions in the comments section below. For more insights into the science of CO2 and related topics, explore our other articles on environmental science and chemistry. Together, we can contribute to a better understanding of the world around us.