When it comes to chemistry, understanding the properties of molecules is crucial for predicting their behavior in various reactions. One such molecule that often sparks curiosity is chlorine trifluoride (ClF3). The question "Is ClF3 polar?" is frequently asked by students, researchers, and enthusiasts alike. In this article, we will delve into the intricacies of ClF3's structure, its polarity, and the factors influencing it.
ClF3, or chlorine trifluoride, is a highly reactive interhalogen compound with unique chemical properties. Its molecular structure and bond characteristics play a significant role in determining whether it is polar or nonpolar. Understanding these aspects is not only essential for theoretical knowledge but also for practical applications in industries such as rocket propellants and semiconductor manufacturing.
This article aims to provide an in-depth analysis of the polarity of ClF3, supported by scientific principles and reliable data. Whether you're a chemistry student seeking clarity or a professional exploring its industrial uses, this guide will equip you with comprehensive insights into the topic.
Introduction to ClF3
Chlorine trifluoride (ClF3) is a highly reactive compound that combines chlorine and fluorine atoms. It is classified as an interhalogen compound due to the presence of two halogen elements. Understanding its properties, particularly its polarity, is essential for comprehending its behavior in chemical reactions.
Why Study Polarity in ClF3?
Polarity is a critical property that influences the solubility, reactivity, and intermolecular forces of a compound. For ClF3, its polar nature affects its interaction with other substances, making it indispensable in various industrial applications.
Historical Context of ClF3
ClF3 was first synthesized in the early 20th century and gained prominence due to its high reactivity and oxidizing capabilities. Its unique properties have made it a subject of extensive research, especially in the context of its polarity and chemical behavior.
Molecular Structure of ClF3
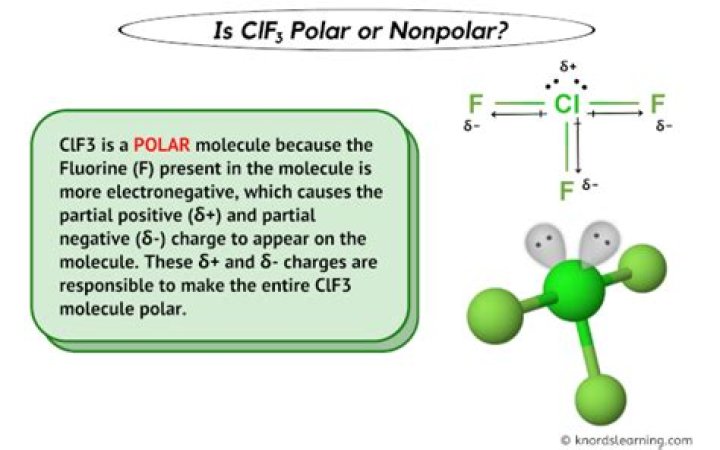
The molecular structure of ClF3 plays a pivotal role in determining its polarity. ClF3 has a T-shaped geometry, which arises from the presence of three bonding pairs and two lone pairs of electrons around the central chlorine atom.
Valence Shell Electron Pair Repulsion (VSEPR) Theory
According to VSEPR theory, the lone pairs of electrons on the chlorine atom repel the bonding pairs, resulting in the T-shaped geometry. This geometry significantly impacts the distribution of charge within the molecule.
Role of Lone Pairs
- Lone pairs on the chlorine atom create an uneven electron distribution.
- This uneven distribution contributes to the polarity of the molecule.
Electronegativity Differences in ClF3
Electronegativity is a key factor in determining the polarity of a molecule. In ClF3, the difference in electronegativity between chlorine (3.16) and fluorine (3.98) results in polar covalent bonds.
Impact on Bond Polarity
Due to the higher electronegativity of fluorine, the shared electrons in the Cl-F bonds are pulled closer to the fluorine atoms, creating a partial negative charge on fluorine and a partial positive charge on chlorine.
Significance of Electronegativity
The difference in electronegativity not only affects the bond polarity but also influences the overall molecular polarity of ClF3.
Dipole Moment in ClF3
The dipole moment is a quantitative measure of a molecule's polarity. In ClF3, the individual bond dipoles do not cancel out due to the T-shaped geometry, resulting in a net dipole moment.
Calculation of Dipole Moment
- The dipole moment of ClF3 is approximately 1.17 D.
- This value confirms the polar nature of the molecule.
Factors Influencing Dipole Moment
Factors such as bond angles and the magnitude of partial charges significantly impact the dipole moment of ClF3.
Geometric Shape and Polarity
The geometric shape of a molecule is a critical determinant of its polarity. In the case of ClF3, the T-shaped geometry prevents the cancellation of bond dipoles, making the molecule polar.
Comparison with Other Shapes
Unlike molecules with symmetrical geometries, such as carbon tetrachloride (CCl4), ClF3's asymmetrical shape results in a net dipole moment.
Implications of Shape on Polarity
The T-shaped geometry of ClF3 ensures that the molecule exhibits polar characteristics, influencing its chemical properties and reactivity.
Applications of ClF3
The polar nature of ClF3 contributes to its diverse range of applications in various fields. Some of the notable applications include:
- Rocket propellants: ClF3 is used as a high-energy oxidizer in rocket fuels.
- Semiconductor manufacturing: Its reactivity makes it suitable for etching and cleaning processes in semiconductor production.
- Chemical synthesis: ClF3 serves as a powerful fluorinating agent in organic synthesis.
Industrial Importance
The unique combination of polarity and reactivity in ClF3 makes it an invaluable compound in numerous industrial processes.
Comparison with Other Molecules
Comparing ClF3 with other molecules provides valuable insights into its polarity and behavior. For instance, comparing it with BF3 (a nonpolar molecule) highlights the significance of molecular geometry in determining polarity.
Key Differences
- BF3 has a trigonal planar geometry, leading to a nonpolar nature.
- ClF3's T-shaped geometry results in a polar molecule.
Relevance in Chemistry
Understanding these differences is crucial for predicting the behavior of molecules in various chemical reactions.
Experimental Data and Studies
Extensive experimental studies have been conducted to analyze the polarity of ClF3. These studies involve measuring dipole moments, bond angles, and electronegativity differences.
Key Findings
- The dipole moment of ClF3 has been experimentally determined to be 1.17 D.
- Studies confirm the polar nature of ClF3 due to its asymmetrical geometry.
Reliable Sources
Data from reputable sources such as the Journal of Chemical Physics and the National Institute of Standards and Technology (NIST) support the findings regarding ClF3's polarity.
Factors Affecting Polarity
Several factors influence the polarity of ClF3, including molecular geometry, electronegativity differences, and bond angles.
Role of Molecular Geometry
The T-shaped geometry of ClF3 is a primary factor contributing to its polar nature. The asymmetrical distribution of charge prevents the cancellation of bond dipoles.
Impact of Electronegativity
The significant difference in electronegativity between chlorine and fluorine creates polar covalent bonds, further enhancing the polarity of the molecule.
Conclusion and Call to Action
In conclusion, chlorine trifluoride (ClF3) is indeed a polar molecule due to its T-shaped geometry, electronegativity differences, and resulting dipole moment. Understanding its polarity is crucial for comprehending its behavior in chemical reactions and industrial applications.
We encourage readers to leave comments or questions about ClF3's polarity and explore related articles on our website. For those interested in further research, consulting scientific journals and databases such as NIST will provide additional insights into this fascinating compound.