Carbon dioxide (CO2) molecular geometry is a fascinating subject that delves into the arrangement of atoms in this vital molecule. CO2 plays a crucial role in our atmosphere, contributing to the greenhouse effect and global climate regulation. Understanding its molecular geometry helps us grasp its behavior and interactions with other substances.
Molecular geometry is the cornerstone of chemistry, offering insights into how molecules behave and function. CO2 is a simple yet significant molecule, composed of one carbon atom and two oxygen atoms. Its linear geometry and symmetric structure make it a perfect candidate for studying molecular behavior.
Through this article, we aim to provide an in-depth analysis of CO2 molecular geometry, covering its structure, properties, and implications in chemistry and environmental science. Whether you're a student, a researcher, or simply curious about the world of molecules, this guide will offer valuable insights into the world of CO2.
Introduction to CO2 Molecular Geometry
The study of CO2 molecular geometry revolves around understanding the spatial arrangement of atoms in the carbon dioxide molecule. Carbon dioxide is a linear molecule, meaning its atoms are aligned in a straight line. This arrangement is dictated by the valence shell electron pair repulsion (VSEPR) theory, which predicts molecular shapes based on electron repulsion.
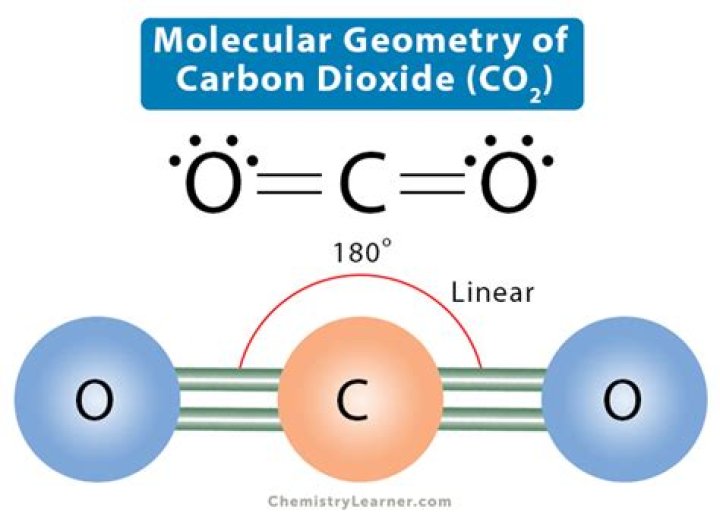
CO2 molecular geometry is linear, with a bond angle of 180 degrees. The molecule's symmetry contributes to its nonpolar nature, making it an excellent solvent for various substances. Furthermore, CO2's geometric structure influences its reactivity and interactions with other molecules.
This section will explore the fundamental principles behind CO2 molecular geometry and why it is essential in chemistry and environmental science.
CO2 Structure and Bonding
Atomic Composition
CO2 consists of one carbon atom and two oxygen atoms. The carbon atom forms double bonds with each oxygen atom, creating a stable and symmetric molecule. This double bonding is crucial in determining the molecule's geometry and properties.
Bonding Mechanism
The bonding in CO2 involves the sharing of electrons between carbon and oxygen atoms. Each oxygen atom shares two electrons with the carbon atom, forming a double bond. This arrangement ensures that all atoms achieve a stable electron configuration, similar to that of noble gases.
Understanding the bonding mechanism in CO2 provides insights into its stability and reactivity. The double bonds contribute to the molecule's rigidity and resistance to chemical reactions.
VSEPR Theory and CO2
Valence Shell Electron Pair Repulsion (VSEPR) theory is a model used to predict the geometry of molecules based on the repulsion between electron pairs. In the case of CO2, the theory predicts a linear geometry due to the absence of lone pairs on the central carbon atom.
The VSEPR model considers the number of electron pairs surrounding the central atom and their spatial arrangement. For CO2, the central carbon atom has two bonding pairs and no lone pairs, resulting in a linear shape with a bond angle of 180 degrees.
This section explains the application of VSEPR theory to CO2 and its implications for molecular geometry.
Linear Geometry of CO2
Characteristics of Linear Geometry
The linear geometry of CO2 is characterized by its straight-line arrangement of atoms and a bond angle of 180 degrees. This geometry arises from the symmetric distribution of electron pairs around the central carbon atom.
Implications of Linear Geometry
The linear geometry of CO2 has significant implications for its physical and chemical properties. It contributes to the molecule's nonpolar nature, making it insoluble in water but soluble in organic solvents. Additionally, the linear shape enhances CO2's stability and resistance to chemical reactions.
This section delves into the characteristics and implications of the linear geometry of CO2.
Symmetry and Properties
Symmetry plays a crucial role in determining the properties of CO2. The molecule's symmetric structure results in a balanced distribution of charge, making it nonpolar. This nonpolar nature influences its interactions with other substances and its behavior in various environments.
Key properties of CO2 include:
- High stability
- Nonpolar nature
- Excellent solvent properties
- Contribution to the greenhouse effect
This section explores the symmetry of CO2 and its impact on the molecule's properties.
Bond Angle in CO2
Understanding Bond Angles
The bond angle in CO2 is 180 degrees, a direct result of its linear geometry. This bond angle arises from the symmetric arrangement of electron pairs around the central carbon atom, minimizing repulsion and maximizing stability.
Significance of Bond Angle
The bond angle in CO2 is significant as it determines the molecule's shape and influences its interactions with other molecules. A bond angle of 180 degrees ensures optimal stability and symmetry, contributing to the molecule's nonpolar nature.
This section examines the bond angle in CO2 and its importance in molecular geometry.
Environmental Impact
CO2 is a major contributor to the greenhouse effect, playing a critical role in regulating Earth's climate. Its molecular geometry and properties make it an effective absorber of infrared radiation, trapping heat in the atmosphere and contributing to global warming.
According to the Intergovernmental Panel on Climate Change (IPCC), CO2 levels have increased significantly over the past century due to human activities such as fossil fuel combustion and deforestation. This rise in CO2 levels has profound implications for global climate patterns and ecosystems.
This section discusses the environmental impact of CO2 and its role in climate change.
Applications in Industry
CO2 finds extensive applications in various industries due to its unique properties. Some of its industrial uses include:
- Carbonation in beverages
- Fire extinguishers
- Refrigerants
- Supercritical CO2 extraction
- Enhanced oil recovery
The linear geometry and nonpolar nature of CO2 make it an ideal candidate for these applications. Its stability and solubility properties further enhance its utility in industrial processes.
This section highlights the diverse applications of CO2 in industry.
Frequently Asked Questions
What is the molecular geometry of CO2?
The molecular geometry of CO2 is linear, with a bond angle of 180 degrees. This geometry arises from the symmetric arrangement of electron pairs around the central carbon atom.
Why is CO2 nonpolar?
CO2 is nonpolar due to its symmetric geometry, which results in a balanced distribution of charge. The equal sharing of electrons between carbon and oxygen atoms ensures that the molecule has no net dipole moment.
How does CO2 contribute to the greenhouse effect?
CO2 absorbs infrared radiation, trapping heat in the Earth's atmosphere and contributing to the greenhouse effect. Its molecular geometry and properties make it an effective absorber of heat, influencing global climate patterns.
This section addresses common questions about CO2 molecular geometry and its implications.
Conclusion
In conclusion, CO2 molecular geometry is a vital subject that offers insights into the structure and behavior of this essential molecule. The linear geometry and symmetric structure of CO2 contribute to its stability, nonpolar nature, and diverse applications in industry and environmental science.
We encourage readers to explore further resources and studies on CO2 molecular geometry. By understanding the science behind CO2, we can better address the challenges posed by climate change and harness the molecule's potential for sustainable development.
Please leave your thoughts and questions in the comments section below. Share this article with others who may find it informative, and explore our other articles for more in-depth knowledge on related topics.