Chlorine trifluoride (ClF3) is a highly reactive and potent chemical compound that has fascinated chemists and researchers for decades. Its unique Lewis structure plays a crucial role in understanding its reactivity and bonding characteristics. If you're curious about the chemistry behind chlorine trifluoride and its Lewis structure, you're in the right place. This article will provide a detailed exploration of ClF3, its structure, properties, and applications.
Understanding the Lewis structure of chlorine trifluoride is essential for anyone interested in advanced chemistry. By delving into its molecular geometry, bond angles, and electron distribution, we can gain insights into its behavior in chemical reactions. Whether you're a student, researcher, or simply a chemistry enthusiast, this article will serve as a valuable resource.
In this guide, we'll cover everything from the basics of Lewis structures to the specific characteristics of ClF3. You'll learn about its molecular geometry, hybridization, and the reasons behind its exceptional reactivity. By the end, you'll have a comprehensive understanding of chlorine trifluoride and its importance in modern chemistry.
Introduction to Chlorine Trifluoride
Chlorine trifluoride (ClF3) is a highly reactive interhalogen compound that consists of one chlorine atom and three fluorine atoms. It was first synthesized in the early 20th century and has since become an important compound in the field of chemistry. ClF3 is primarily used in the production of uranium hexafluoride (UF6) for nuclear fuel processing.
Its high reactivity makes chlorine trifluoride both useful and dangerous. In its gaseous state, ClF3 is colorless, but it can react violently with water, metals, and organic materials. This makes it essential to handle with extreme care in laboratory and industrial settings.
Chemical Properties of ClF3
Here are some key chemical properties of chlorine trifluoride:
- Molecular formula: ClF3
- Molar mass: 92.45 g/mol
- Melting point: -76°C (-104.8°F)
- Boiling point: 11.75°C (53.15°F)
Lewis Structure Basics
A Lewis structure is a diagram that represents the bonding between atoms in a molecule and the lone pairs of electrons. It is a fundamental concept in chemistry that helps explain molecular geometry, hybridization, and reactivity. Understanding Lewis structures is crucial for predicting the behavior of chemical compounds.
When drawing a Lewis structure, the goal is to achieve a stable electron configuration for each atom in the molecule. This typically involves filling the outermost electron shells of the atoms with eight electrons (the octet rule), although there are exceptions for certain elements like hydrogen and helium.
Chlorine Trifluoride Lewis Structure
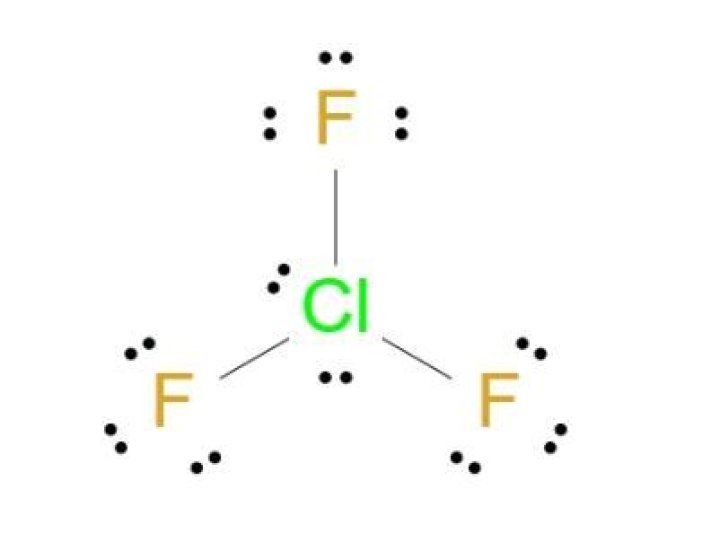
The Lewis structure of chlorine trifluoride involves one chlorine atom bonded to three fluorine atoms. Chlorine, being the central atom, forms single covalent bonds with each fluorine atom. Additionally, chlorine has two lone pairs of electrons, making the total number of electron pairs around the central atom five.
Here’s a breakdown of the chlorine trifluoride Lewis structure:
- Central atom: Chlorine (Cl)
- Peripheral atoms: Three fluorine (F) atoms
- Lone pairs: Two lone pairs on the chlorine atom
Steps to Draw the ClF3 Lewis Structure
Follow these steps to draw the Lewis structure of chlorine trifluoride:
- Count the total number of valence electrons: Chlorine has 7 valence electrons, and each fluorine atom contributes 7 electrons. Total = 28 electrons.
- Place the chlorine atom at the center and surround it with three fluorine atoms.
- Form single bonds between chlorine and each fluorine atom, using 6 electrons.
- Complete the octets of the fluorine atoms by adding lone pairs, using 18 electrons.
- Place the remaining two lone pairs on the chlorine atom.
Molecular Geometry of ClF3
The molecular geometry of chlorine trifluoride is T-shaped. This geometry arises due to the presence of two lone pairs of electrons on the central chlorine atom. According to the VSEPR (Valence Shell Electron Pair Repulsion) theory, lone pairs repel bonding pairs more strongly than bonding pairs repel each other. As a result, the three bonding pairs are pushed into a T-shaped arrangement.
Factors Influencing Molecular Geometry
Several factors influence the molecular geometry of ClF3:
- Number of bonding pairs: Three
- Number of lone pairs: Two
- Electron pair geometry: Trigonal bipyramidal
- Molecular geometry: T-shaped
Hybridization in Chlorine Trifluoride
The hybridization of chlorine in ClF3 is sp3d. This hybridization occurs because chlorine uses five orbitals to form bonds with the three fluorine atoms and accommodate the two lone pairs. The sp3d hybridization results in a trigonal bipyramidal electron geometry, which aligns with the observed T-shaped molecular geometry.
Importance of Hybridization
Hybridization plays a critical role in determining the shape and reactivity of molecules. In the case of chlorine trifluoride, the sp3d hybridization explains the distribution of electron pairs and the resulting molecular geometry. This understanding is essential for predicting how ClF3 will interact with other substances.
Bond Angles in ClF3
The bond angles in chlorine trifluoride are slightly less than 90° and 180° due to the repulsion between lone pairs and bonding pairs of electrons. Specifically:
- The angle between the axial fluorine atoms and the equatorial fluorine atom is approximately 87.5°.
- The angle between the two axial fluorine atoms is approximately 175°.
These deviations from ideal bond angles are a result of lone pair-bond pair repulsion, as predicted by VSEPR theory.
Electronegativity and Polarity
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. Fluorine is the most electronegative element, with a value of 4.0 on the Pauling scale, while chlorine has an electronegativity of 3.0. This difference in electronegativity makes the Cl-F bonds polar, with the fluorine atoms pulling electron density away from the chlorine atom.
Despite the polar bonds, chlorine trifluoride is a nonpolar molecule overall due to its symmetrical T-shaped geometry. The bond dipoles cancel out, resulting in a net dipole moment of zero.
Reactivity of Chlorine Trifluoride
Chlorine trifluoride is one of the most reactive chemicals known. It reacts violently with water, metals, and organic materials, often producing hazardous byproducts such as hydrogen fluoride (HF) and chlorine gas (Cl2). Its high reactivity stems from the strong oxidizing and fluorinating properties of ClF3.
Here are some examples of reactions involving chlorine trifluoride:
- Reaction with water: ClF3 + 2H2O → HF + HClO + O2
- Reaction with metals: ClF3 + 2Fe → FeCl3 + FeF3
- Reaction with organic compounds: ClF3 + CxHy → CFx + HF
Applications of ClF3
Despite its hazardous nature, chlorine trifluoride has several important applications in industry and research:
- Nuclear fuel processing: ClF3 is used to produce uranium hexafluoride (UF6), a key material in the enrichment of uranium for nuclear reactors.
- Semiconductor manufacturing: It serves as an etchant in the production of integrated circuits and microchips.
- Propellants: ClF3 has been investigated for use in rocket propellants due to its high oxidizing capacity.
Safety Precautions
Handling chlorine trifluoride requires extreme caution due to its high reactivity and potential for violent reactions. Here are some safety precautions to follow:
- Use proper personal protective equipment (PPE), including gloves, goggles, and a respirator.
- Store ClF3 in sealed containers away from water, metals, and organic materials.
- Work in a well-ventilated area or fume hood to avoid inhalation of toxic vapors.
- Dispose of ClF3 according to local regulations and guidelines.
Conclusion
Chlorine trifluoride is a fascinating and powerful chemical compound with a unique Lewis structure and molecular geometry. Understanding its properties, reactivity, and applications is essential for anyone working in advanced chemistry or related fields. By following proper safety precautions, researchers and industry professionals can harness the potential of ClF3 while minimizing risks.
We encourage you to leave a comment or share this article if you found it informative. For further reading, explore our other articles on chemistry and related topics. Stay curious and keep learning!